Abstract
To determine the biological and clinical relevance of programmed death 1 (PD-1) in follicular lymphoma (FL), we characterized PD-1+ T-cell subsets and assessed their biological function as well as potential clinical impact. We found that PD-1 is expressed on intratumoral CD4+ T cells with both bright and dim intensity, representing two different sub-populations of cells. By immunohistochemistry, we found that CD4+PD-1high T cells predominantly reside in the lymph node follicles, while PD-1low T cells are mainly located in an interfollicular pattern. Intratumoral CD4+PD-1high T cells have a TFH cell phenotype, express CXCR5, secrete IL-21 and are BCL-6 positive with no TIM-3 expression. In contrast, CD4+PD-1low T cells have an exhausted phenotype, express TIM-3 and do not express BCL-6 and CXCR5. Functionally, CD4+PD-1high T cells actively supported B-cell growth, while CD4+PD-1low T cells displayed a reduced cytokine production and cell-signal transduction. Clinically, we observed that the numbers of CD4+ or CD8+PD-1low T cells significantly correlate with a reduced overall survival in FL patients (P=0.007 and 0.04 respectively; n=32). In contrast, the number of CD4+PD-1high T cells was not associated with patient outcome. Taken together, these results indicated that PD-1 expression defines two sub-populations with distinct functions that differentially impact patient outcome in FL.
Similar content being viewed by others
Introduction
Follicular lymphoma (FL) is characterized by the presence of a significant number of T cells in the tumor microenvironment that have a substantial impact on anti-tumor immunity and patient outcome.1, 2 Previous studies have shown that the type of T-cell-mediated immune response determines the anti-tumor immunity3, 4, 5 thereby impacting patient outcome.6, 7, 8 In FL, lymph node follicles remain intact, while the germinal centers become larger due to increasing numbers of lymphoma B cells. Similar to normal lymph nodes, T cells reside both outside and inside the follicles. These intratumoral T cells are generally heterogenous and influence tumor immunity both positively and negatively, depending on the prevalence of various T-cell populations and location of the cells. For example, while high numbers of tumor-infiltrating Foxp3+ cells are associated with an improved overall survival in FL,8 patients with Foxp3+ cells located in a follicular pattern (intrafollicular or perifollicular) had a significantly elevated risk of transformation and shorter survival than patients with Foxp3+ cells present in a diffuse pattern.9
Programmed death 1 (PD-1), a member of the CD28/CTLA-4 family, has been shown to negatively regulate T-cell receptor signaling and decrease proliferation and cytokine production in T cells. The involvement of regulatory T cells (Treg) in the interaction of PD-1 and PD-L1 contributes to PD-1-mediated inhibition of T cells. Recently, PD-1 has been demonstrated to be a major component in the process of T-cell exhaustion,10, 11 although complex mechanisms are involved in inducing exhaustion. It has been shown that TIM-3, a family member of T-cell immunoglobulin and mucin domain proteins, along with other molecules, are co-expressed with PD-1 on exhausted T cells.12, 13 In contrast however, PD-1 is also expressed on T cells present within the follicles of secondary lymphoid organs, and this subset of T cells has been identified as follicular helper T (TFH) cells.14, 15, 16 Because of the unique location, TFH cells are able to closely interact with B cells to promote the formation of germinal centers and to help B cells produce long-lived memory B cells and high-affinity antibodies.
In a previous work, we observed that PD-1 was expressed by different subsets of intratumoral T cells in FL including TFH cells and exhausted T effector cells.17, 18 However, the function, prevalence, distribution and clinical importance of the respective intratumoral PD-1+ T subsets in FL patients are largely unknown. In this study, we determined the expression of PD-1 and identified the location and intensity of PD-1 staining in the tumor tissue of FL by immunohistochemistry and flow cytometry. We measured the biological function of different subsets of PD-1+ T cells and clinical impact in FL patients.
Materials and methods
Patient samples
Patients providing written informed consent were eligible for this study if they had a tissue biopsy that on pathologic review showed follicular B-cell non-Hodgkin's lymphoma and adequate tissue or peripheral blood to perform the experiments. Peripheral blood mononuclear cells from healthy donors and lymph nodes from patients with hyperplasia were used as controls. The use of human tissue samples for this study was approved by the Institutional Review Board of the Mayo Clinic/Mayo Foundation.
Cell isolation and purification
Fresh tumor biopsy specimens from patients with FL and normal lymph nodes were gently minced over a wire mesh screen to obtain a cell suspension. The cell suspension or peripheral blood from patients or healthy donors was centrifuged over Ficoll Hypaque at 500 g for 15 min to isolate mononuclear cells. CD3+ T cells, CD14+ monocytes or CD19+ B cells were isolated by using positive selection with CD3, CD14 or CD19 microbeads (StemCell Technologies, Vancouver, BC, Canada). CD3+TIM-3+ or TIM-3− T cells were isolated by CD3 negative selection and the resulting CD3+ T cells were incubated with biotin-conjugated TIM-3 antibody followed by incubation with streptavidin-conjugated microbeads
Cell coculture and viability assay
CXCR5-depleted CD4+ T cells were obtained by CD4 negative selection and the resulting CD4+ T cells were incubated with biotin-conjugated CXCR5 antibody followed by incubation with streptavidin-conjugated microbeads. Lymphoma cells were purified by CD19 positive selection. CXCR5-depleted or CXCR5-undepleted CD4+ T cells were co-cultured with CD19+ lymphoma B cells in the presence or absence of CD40L (100 ng/ml) or LPS (1 μg/ml) for 3 days. Annexin V and propridium iodide staining were performed to measure the viability of CD19+ lymphoma B cells.
Intracellular staining and flow cytometry
For profiling of cytokine production by PD-1highCXCR5+ or PD-1lowTIM-3+ T cells, fresh-isolated mononuclear cells were stimulated with phorbol myristate acetate and ionomycin in the presence of a protein transport inhibitor Brefeldin A for 5 h. After fixation and permeabilization, cells were stained with fluorochrome-conjugated antibodies for IL-2, IFN-γ or IL-21 plus surface marker antibodies for CD4, TIM-3 or CXCR5 in each specimen. Cells were then analyzed on a flow cytometer.
Transcriptional factor Foxp3 expression detection
Foxp3 and Bcl-6 expression was determined by flow-based intracellular staining following the manufacturer’s instructions. Cells were fixed and permeabilized with reagents from a Foxp3-staining kit (BioLegend). Cells were then stained with fluorochrome-conjugated Abs against Foxp3 or Bcl-6 plus fluorochrome-conjugated anti-CD4, PD-1 and TIM-3 or CXCR5 Abs for 30 min and analyzed by flow cytometry.
Phosphorylation assay
The phosphorylation of STATs was detected following the manufacturer’s instructions (BD Biosciences, San Jose, CA, USA). Briefly, fresh-isolated MNCs stimulated with or without phorbol myristate acetate/ionomycin or cytokines for 30 min and then fixed and permeabilize by using a phosflow kit (BD Biosciences). Cells were stained with anti-Stat1, Stat3, Stat4 or Stat5-Alexa647 antibody plus anti-CD3-FITC and TIM-3-PE antibodies for 30 min and analyzed by flow cytometry.
Immunohistochemistry
Paraffin embedded tissue was obtained from Mayo Clinic Tissue Registry and cut serially at 5 μm. The tissue sections were deparaffinized in three changes of xylene and cleared through graded ethanol series. Endogenous peroxidase was quenched by incubation in 50% methanol/H2O2. After rinsing with tap water, all sections were pretreated 30 min with 50 mM EDTA, pH 8.0 using a steamer and cooled for an additional 5 min. All immunohistochemical staining was performed automatically on DAKO Autostainerplus using the following antibodies and their corresponding detection systems: PD-1 (Abcam, 1 mg/ml, ab#52587, 1:50); TIM-3 (R&D, AF2365, 1:200); CXCR5 (Abcam, #ab46218, 1:100); or mouse IgG1 control (DAKO, #x0931, 1:100000). All sections were stained with hematoxylin and rinsed well in tap water. All slides were observed with light microscopy (Olympus AX70, 200 x/aperture 0.46, 400 x/aperture 0.75, 600 × /aperture 0.80; Olympus America, Melville, NY, USA) with images captured with a SPOT RT camera and software (Diagnostic Instruments, Burlingame, CA, USA).
Statistical analysis
Statistical analysis was performed by using Student's t test. Significance was determined at P<0.05. Overall survival was measured from the date of diagnosis until death from any cause. Patients alive and still at risk of death at last follow-up evaluation were censored for the analysis of overall survival. Survival of all patients was estimated by using the Kaplan–Meier method. The univariate association between PD-1 expression and survival was determined with the log-rank test.
Results
PD-1 is expressed in the tumor microenvironment of FL
It has been shown that signaling through PD-1 has a critical role in T-cell-mediated immune responses in a variety of pathophysiological conditions. To determine the role of PD-1 in FL, we first measured its expression in biopsy lymph nodes of FL patients. By immunohistochemistry, we observed that PD-1 was highly expressed in FL biopsy specimens (Figure 1a). By flow cytometry, we found that PD-1 was abundantly expressed on intratumoral CD3+ T cells while CD19+ B cells and CD14+ monocytes had a negligible expression of PD-1 on cell surface (Figure 1b).
PD-1 is abundantly expressed in the tumor microenvironment of FL. (a) A representative image showing PD-1 expression determined by immunohistochemistry on a tissue section from a FL patient. Image of isotype Ab was stained for control to PD-1 Ab, n=4. (b) Representative plots showing PD-1 expression on CD3+, CD19+ or CD14+ cells from biopsy specimens of a FL patient, n=10. (c) Representative plots showing PD-1 expression on CD4+ or CD8+ T cells from biopsy specimens of FL. PD-1 expression was divided into PD-1high and PD-1low, n=10. (d) Representative images showing PD-1 expression determined by immunohistochemistry on a tissue slide from a FL patient. PD-1 staining can be grouped into PD-1bright and PD-1dim, n=4.
Next, we determined PD-1 expression on intratumoral CD4+ and CD8+ T cells in FL. We saw a different pattern of PD-1 expression between the CD4+ and CD8+ subset. For CD4+ T cells, we could distinguish CD4+ T cells as two populations based on PD-1 expression (Figure 1c). Some CD4+ T cells had PD-1 expressed at high levels (PD-1high), while other cells expressed a low level of PD-1 (PD-1low). For CD8+ T cells, the PD-1high population was not seen and only the PD-1low population existed (Figure 1c). Supporting this finding, we observed that PD-1 stained brightly in cells mostly within the follicles (Figure 1d). However, at high power, cells that stained dimly for PD-1 were seen in the interfollicular region (Figure 1d).
CD4+PD-1high T cells express CXCR5 in FL
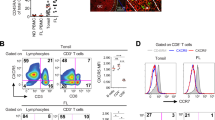
Based on PD-1 expression levels, it appeared that CD4+PD-1+ T cells are heterogeneous and could be divided into two subsets, PD-1high and PD-1low. Recent studies have identified a CD4+PD-1+ T-cell population in the germinal center of follicles in secondary lymphoid organs as follicular T helper (TFH) cells with co-expression of CXCR5. Therefore, we determined co-expression of PD-1 and CXCR5 on intratumoral CD4+ T cells. As shown in Figure 2a, CXCR5 was highly expressed on a subset of CD4+ T cells, accounting for ~46% of the CD4+ T cells. The percentage of CXCR5-expressing CD4+ T cells varied in a range from 13.5 to 80.9% (n=28), suggesting variability in the number of CD4+ T cells residing in the follicles among patients. For CD8+ T cells, CXCR5 expression was negligible and <7% of CD8+ T cells expressed CXCR5. Next, we tested which intratumoral CD4+ subset expressed CXCR5. We found that CXCR5 was predominantly expressed on the surface of CD4+PD-1high T cells, and ~70% of CD4+PD-1high T cells expressed CXCR5. In contrast, we observed that the intratumoral CD8+ T cells lacked the PD-1high population and there was minimal CXCR5 expression on CD8+ cells (Figure 2b).
CD4+PD-1high T cells express CXCR5 in FL. (a) Representative dot plots showing CXCR5 expression on CD4+ or CD8+ T cells from lymph nodes of a FL patient. The right graph summarizes percentages of CXCR5+ cells in CD4+ or CD8+ T subset in FL, n=32. (b) Representative dot plots showing CXCR5 and PD-1 expression on CD4+ or CD8+ T cells from biopsy specimens of a FL patient. The plots were pre-gated on CD4+ or CD8+ cells. The right is a graph summarizing percentages of CXCR5+ cells in CD4+PD-1high sub-population in FL, n=32. (c) Representative image showing PD-1 or CXCR5 expression determined by immunohistochemistry on tissue sections from a FL patient. (d) Representative histograms showing TIM-3, 2B4, LAG-3, CD70 and OX40 expression on intratumoral CD4+PD-1high T cells from FL.
To identify PD-1high and CXCR5+ cells within FL tissue, we stained tissue sections for PD-1 and CXCR5 (Figure 2c). Again, we saw bright staining of PD-1 on cells in the follicular areas. Concomitant with the bright PD-1 expression pattern, CXCR5 is also expressed on cells in the similar area of tumor tissue in FL (Figure 2c), suggesting a co-expression pattern between high PD-1 expression and CXCR5 expression on cells in the follicles.
We next determined whether CD4+PD-1high T cells in FL had any evidence of an exhausted phenotype. As shown in Figure 2d, CD4+PD-1high T cells lack expression of exhaustion markers such as TIM-3, 2B4 and LAG-3 but expressed co-stimulatory molecules including CD70 and OX40.
CXCR5-expressing CD4+PD-1high T cells in FL are follicular T helper cells
It has been shown that BCL-6 is a master transcriptional factor and lineage marker of TFH cells. Therefore, to further characterize CXCR5-expressing CD4+PD-1high T cells, we measured BCL-6 expression in intratumoral CD4+PD-1high T cells in FL. As shown in Figure 3a, CD4+PD-1high T cells expressed high levels of BCL-6 compared with CD4+PD-1low or CD4+PD-1neg T cells. Similarly, IL-21, a cytokine mainly secreted by TFH cells, was highly expressed by CD4+PD-1high T cells compared with CD4+PD-1low or CD4+PD-1neg T cells (Figure 3a).
CXCR5-expressing CD4+PD-1high T cells are follicular T helper cells in FL. (a) Representative histograms showing expression of BCL-6 or IL-21 in CD4+PD-1high or PD-1low cells from biopsy specimens of FL patients. PD-1high or PD-1low subset was defined and gated on PD-1 expression levels shown in a plot (left). (b) Representative dot plots showing co-staining of annexin V and propidium iodide (PI) in CD19+ lymphoma B cells co-cultured with CXCR5-depleted (−) or CXCR5-undepleted (+) CD4+ T cells for 3 days. Below: Graph summarizing viability of CD19+ lymphoma B cells co-cultured with CXCR5-depleted (−) or CXCR5-undepleted (+) CD4+ T cells in the presence or absence of CD40L or LPS for 3 days. Cell viability was measured using annexin V and PI staining and reported as the numbers of cells lacking annexin V and PI staining (n=6).
Because of the proximity between CD4+PD-1high and lymphoma B cells in FL, we next wanted to know whether CD4+PD-1high T cells have impact on lymphoma cell survival. To do this, we co-cultured CD19+ lymphoma B cells with CXCR5-depleted or CXCR5-undepleted CD4+ T cells in the presence or absence of CD40L or LPS for 3 days. The viability of lymphoma B cells was measured by annexin V and propridium iodide staining. As shown in Figure 3b, the viability of lymphoma cells was significantly greater when co-cultured with CXCR5-undepleted CD4+ T cells compared with those co-cultured with CXCR5-depleted CD4+ T cells, as indicated by an increased number of viable (annexin V-PI-) cells in the group of CXCR5-undepleted CD4+ T cells compared with the group of CXCR5-depleted CD4+ T cells. Similar results were seen when normal TFH cells from normal tonsil were co-cultured with normal tonsillar B-cells, suggesting that TFH cells support the viability of B-cells in general and do not specifically support lymphoma B-cell growth. Taking all data together, these results indicated that CD4+PD-1high T cells are TFH cells.
CD4+PD-1low T cells express TIM-3 in FL
We next wanted to phenotypically characterize the CD4+PD-1low T-cell subset. It has been shown that TIM-3 is involved in the induction of T-cell exhaustion and is co-expressed with PD-1 on exhausted T cells. Therefore, we first determined TIM-3 expression on intratumoral CD4+ T cells. As shown in Figure 4a, TIM-3 was abundantly expressed on a subset of both CD4+ and CD8+ T cells, which accounted for ~30% (range: 16.2–68.1%, n=25) of CD4+ and 40% (range: 9.2–85.3%, n=25) of CD8+ T cells in FL, respectively. Next, we examined which intratumoral CD4+ subset expressed TIM-3. We found that TIM-3 was predominantly expressed on the CD4+PD-1low T-cell subset while CD4+PD-1high T cells had negligible expression of TIM-3 (Figure 4b). Approximately 40% of CD4+PD-1low T cells expressed TIM-3. Furthermore, we observed that the intratumoral CD8+ T-cell subset that were PD-1low, also expressed high levels of TIM-3 on their surface, and TIM-3+ cells accounted for 45% of CD8+PD-1low T cells (Figure 4b).
CD4+PD-1low T cells express TIM-3 in FL. (a) Representative dot plots showing TIM-3 expression on CD4+ or CD8+ T cells from lymph nodes of a FL patient. The right graph summarizes percentages of TIM-3+ cells in CD4+ or CD8+ T subset in FL, n=32. (b) Representative dot plots showing TIM-3 and PD-1 expression on CD4+ or CD8+ T cells from biopsy specimens of a FL patient. The plots were pre-gated on CD4+ or CD8+ cells. The right is a graph summarizing percentages of TIM-3+ cells in CD4+ or CD8+PD-1low sub-population in FL, n=32. (c) Representative image showing PD-1 or TIM-3 expression determined by immunohistochemistry on tissue sections from a FL patient. (d) Representative histograms showing TIM-3, 2B4, LAG-3, CD70 and OX40 expression on intratumoral CD4+PD-1low or CD8+PD-1low T cells from B-cell non-Hodgkin's lymphoma.
Next, to topographically identify PD-1low and TIM-3+ cells in FL tissue, we stained tissue sections for PD-1 and TIM-3. We again saw bright staining for PD-1 on cells in the follicles, while weak staining for PD-1 could be seen between follicles. Similar to the PD-1low expression pattern, TIM-3 was expressed on cells between the follicles, suggesting a co-expression pattern between PD-1low and TIM-3 on cells in interfollicular regions (Figure 4c).
Because co-expression of PD-1 and TIM-3 suggested a possible exhausted phenotype, we further tested for additional markers of T-cell activation and exhaustion. As shown in Figure 4d, contrary to CD4+PD-1high T cells, CD4+PD-1low T cells expressed high levels of TIM-3. 2B4, a marker for exhausted T cells, was highly expressed on CD8+PD-1low T cells. Furthermore, CD8+PD-1low T cells lacked expression of co-stimulatory molecule OX40.
CD4+PD-1lowTIM-3+ T cells in FL are functionally exhausted
To functionally characterize PD-1lowTIM-3+ T cells, we first measured cytokine production by this subset using cells from FL specimens. An intracellular staining assay was employed to measure the cytokine production of PD-1lowTIM-3+ T cells stimulated with phorbol myristate acetate/ionomycin in the presence of Brefeldin A for 4 h. As shown in Figure 5a, the vast majority of IL-2- or IFN-γ-producing cells came from TIM-3− T cells. In contrast, PD-1low TIM-3+ T cells failed to produce cytokines such as IL-2 and IFN-γ in both tissue and peripheral blood of FL (Figure 5a). Next, we determined whether cell-signal transduction was also decreased in PD-llowTIM-3+ T cells. Intratumoral T cells from FL were treated with or without IFN-γ, IL-6, IL-12 or IL-2, respectively, and phosphorylation of Stat1, 3, 4 or 5 in PD-1lowTIM-3− or PD-1lowTIM-3+ T cells was measured by Phosflow (BD Bioscience). We found that intratumoral PD-1lowTIM-3+ T cells had reduced cell signaling. Significantly less phosphorylation of Stat1, 3, 4 (indicated by either MFI or % of positive cells from Phosflow staining) was seen in TIM-3+ T cells compared with TIM-3− T cells in response to cytokine stimulation (Figure 5b). Taken together, these results suggest that intratumoral PD-1lowTIM-3+ T cells in FL are functionally exhausted.
CD4+PD-1lowTIM-3+ T cells are functionally exhausted in FL. (a) Representative dot plots showing IL-2 and IFN-γ expression by PD-1lowTIM-3- or PD-1lowTIM-3+ in CD4+ T cells from peripheral blood of healthy donors or biopsy specimens of FL patients. Cytokine production was measured by intracellular staining in intratumoral T cells stimulated with phorbol myristate acetate/ionomycin in the presence of Brefeldin A. (b) Representative histograms showing phosphorylation of Stats in PD-1lowTIM-3+ or PD-1lowTIM-3-T cells in FL. Freshly isolated mononuclear cells were treated with or without IFN-γ, IL-6, IL-12 or IL-2 and phosphorylation of Stat1, 3, 4 or 5 was measured by Phosflow staining. Plots shown were gated on CD3+ cells.
PD-1high or PD-1low T cells predict a poor outcome in FL patients
Next, we wondered whether the presence of PD-1high or PD-1low T cells in the tumor microenvironment has an impact on patient outcomes in FL. To do this, we evaluated a cohort of 32 previously untreated FL patients who had diagnostic biopsy specimens available for analysis and who had been followed for long-term outcome (Table 1). All patients subsequently received chemotherapy in combination with rituximab as therapy for the disease although many patients were initially observed. We first measured the numbers of PD-1+, PD-1highCXCR5+ or PD-1lowTIM-3+ T cells in both CD4 and CD8 population of biopsy specimens from this cohort of patients. On average, the numbers of PD+ cells accounted for 53.13%±18.57% (range: 17.22–92.57%, n=32) and 46.82%±14.71% (range: 12.78–81.73%, n=32) of CD4+ and CD8+ T cells in LNs of FL patients, respectively (Figure 6a). Among CD4+PD-1+ T cells, the numbers of PD-1highCXCR5+ or PD-1lowTIM-3+ T cells were similar and accounted for 26.60%±15.82% and 26.39%±9.76% of CD4+ T cells in LNs of FL patients, respectively. Among CD8+PD-1+ T cells, however, the numbers of PD-1high CXCR5+ were very low (4.83%±7.91%) and PD-1lowTIM-3+ (42.08%±11.88%) accounted for the most of CD8+PD-1+ T cells (Figure 6a).
PD-1high or PD-1low T cells predict transformation and a poor outcome in FL patients. (a) A graph showing percentages of PD-1+, PD-1high or PD-1low cells in CD4+ (left) or CD8+ (right) T population from biopsy specimens of FL patients. The frequency of CD4+PD-1+ or CD8+PD-1+ T cells was determined by flow cytometry (n=32). (b) Kaplan–Meier curves for overall survival (OS) of FL patients (n=32) by the number of CD4+PD-1low (left) or CD8+PD-1low (right) T cells with a cutoff of 26 or 45%, respectively. (c) Kaplan–Meier curves for OS of FL patients (n=32) by the number of CD4+PD-1high T cells with a cutoff of 25%. The numbers of subsets represent the frequency in the total mononuclear cell population from FL specimens.
Given that PD-1 expression levels separated CD4+ T cells into 2 subsets with distinct phenotype and function, we predicted that these two subsets would have a different role in impacting patient outcomes. We therefore correlated the numbers of CD4+PD-1high or CD4+PD-1low T cells with patient survival. As shown in Figure 6b, using 26% as a cutoff point (the mean number of PD-1+CD4+ T cells), we found that the numbers of CD4+PD-1low T cells were associated with a poorer survival in this FL patient cohort (Figure 6b). Similarly, the frequency of CD8+PD-1low T cells also significantly correlated with an inferior outcome in this cohort of patients (Figure 6b). In contrast, we found that the frequency of CD4+PD-1high T cells did not correlated with outcome in FL patients (Figure 6c).
Discussion
The significant finding in the present study is that PD-1 expression levels divide intratumoral T cells into subsets with different phenotype and function that differentially impact outcome of FL patients. These findings may explain why the contribution of intratumoral PD-1+ T cells to patient outcome in FL has been controversial. Although some studies have found that increased numbers of PD-1+ T cells correlate with an inferior prognosis19 or have no impact on patient outcome,20 other studies observed that the numbers of intratumoral PD-1+ T cells are an indicator of a favorable outcome in FL.21 The different PD-1+ cell populations we show to be present in FL may explain the differences in the findings of these studies. Because immunohistochemistry was the major method to count PD-1 positive-staining cells in tissue slides in these studies, this method may count different populations of cells. In the present study, we show that two distinct populations of cells express PD-1 and the prevalence of these subsets may impact patient outcome.
PD-1, a member of the CD28/CTLA-4 family, has been shown to negatively regulate T-cell receptor signaling and decrease proliferation and cytokine production in T cells. However, recent studies demonstrated that PD-1 expression is involved in different functions in T-cell subsets depending on the location where the cells reside. For example, PD-1-expressing T cells located in the follicles are TFH cells that exhibit help function to B cells, which is different from the cells originally described. Our results are in agreement with this finding and we found that a subset of T cells located within follicles that express high levels of PD-1 as well as CXCR5. Similar to previous reports, these CD4+PD-1high cells express Bcl-622 and secret IL-21,23 two molecules specifically expressed by TFH cells.
In addition to its expression on TFH cells, recent findings have shown that PD-1 can be upregulated in exhausted T cells.24 We therefore addressed the question as to which characteristics separate PD-1+ TFH from PD-1+-exhausted T cells. We used FL as a model as both populations are present in FL lymph nodes, and found that PD-1 expression level and location as well as co-expression of CXCR5 or TIM-3 distinguish TFH from exhausted T cells. In FL, TFH cells express high levels of PD-1, reside in intrafollicular regions and co-express CXCR5. In contrast, exhausted T cells express low levels of PD-1, reside in interfollicular regions and co-express TIM-3. CD8+ T cells only have a PD-1low population, in which TIM-3 is co-expressed, suggesting an exhausted phenotype. In a previous report, however, it was found that the PD-1high population contained both TFH and non-TFH T cells and some cells displayed functional changes similar to exhausted T cells including a reduced cytokine signaling.25 In our study, we did not find that PD-1highCXCR5+ T cells are exhausted, but rather that PD-1highCXCR5+ T cells are TFH cells. This is in agreement with the work of others25 who found that functional PD-1highCXCR5+ TFH cells are present in FL and these cells actively secrete IL-21 and IL-4.
Functionally, the CD4+PD-1+ cells also represent two distinct populations. CD4+PD-1high T cells secrete IL-21 and were able to support the survival of lymphoma B cells in vitro, consistent with the finding that TFH cells support B cells. CD4+PD-1low T cells expressed TIM-3 and displayed a reduced cytokine production with a little signal transduction in response to cytokine stimulation. These findings are in agreement with the previous reports that PD-1+TIM-3+ T cells are exhausted and have reduced the capacity to proliferate and produce cytokines.17, 26 Further supporting these findings, we found that PD-1low T cells, especially PD-1lowCD8+ subset, constitutively expressed high level of 2B4, a marker of late stage exhaustion, and lacked expression of OX40, a co-stimulatory molecule.
Clinically, we found that the numbers of both CD4+ and CD8+PD-1low T cells significantly correlated with a poor prognosis in FL patients, which is in line with a previous report that the frequency of intratumoral CD4+TIM-3+ T cells predicts an inferior survival in FL patients. In contrast, the numbers of CD4+PD-1high were not associated with survival in FL patients. Supporting this finding, we observed that patients with transformation had increased PD-1low and decreased PD-1high T cells in FL. These results indicate that PD-1 plays a role not only in T-cell-mediated tumor immunity, but also in tumor cell biology in FL.
In conclusion, we have defined two distinct T-cell sub-populations based on PD-1 expression levels. They are PD-1highCXCR5+ TFH cells and PD-1lowTIM-3+ exhausted T cells. PD-1highCXCR5+ TFH cells are unique to the CD4+ population, while PD-1lowTIM-3+ exhausted T cells can exist in both CD4+ and CD8+ T populations. Topographically, PD-1highCXCR5+ TFH cells are located in intrafollicular regions and PD-1lowTIM-3+ exhausted T cells reside predominantly in interfollicular areas. Functionally, PD-1highCXCR5+ TFH cells support the survival of B cells, but PD-1lowTIM-3+ exhausted T cells have lost the capacity to produce cytokines and transduce cell signaling. Clinically, PD-1highCXCR5+ TFH cells are not associated with patient survival, but PD-1lowTIM-3+ exhausted T cells are associated with a poor outcome in FL patients.
References
Dave SS, Wright G, Tan B, Rosenwald A, Gascoyne RD, Chan WC et al. Prediction of survival in follicular lymphoma based on molecular features of tumor-infiltrating immune cells. New Engl J Med 2004; 351: 2159–2169.
Glas AM, Knoops L, Delahaye L, Kersten MJ, Kibbelaar RE, Wessels LA et al. Gene-expression and immunohistochemical study of specific T-cell subsets and accessory cell types in the transformation and prognosis of follicular lymphoma. J Clin Oncol 2007; 25: 390–398.
Yang ZZ, Novak AJ, Stenson MJ, Witzig TE, Ansell SM . Intratumoral CD4+CD25+ regulatory T-cell-mediated suppression of infiltrating CD4+ T cells in B-cell non-Hodgkin lymphoma. Blood 2006; 107: 3639–3646.
Yang ZZ, Novak AJ, Ziesmer SC, Witzig TE, Ansell SM . Attenuation of CD8(+) T-cell function by CD4(+)CD25(+) regulatory T cells in B-cell non-Hodgkin's lymphoma. Cancer Res 2006; 66: 10145–10152.
Hilchey SP, De A, Rimsza LM, Bankert RB, Bernstein SH . Follicular lymphoma intratumoral CD4+CD25+GITR+ regulatory T cells potently suppress CD3/CD28-costimulated autologous and allogeneic CD8+CD25- and CD4+CD25- T cells. J Immunol 2007; 178: 4051–4061.
Labidi SI, Menetrier-Caux C, Chabaud S, Chassagne C, Sebban C, Gargi T et al. Serum cytokines in follicular lymphoma. Correlation of TGF-beta and VEGF with survival. Ann Hematol 2010; 89: 9.
Jones EA, Pringle JH, Angel CA, Rees RC . Th1/Th2 cytokine expression and its relationship with tumor growth in B cell non-Hodgkin's lymphoma (NHL). Leuk Lymphoma 2002; 43: 1313–1321.
Carreras J, Lopez-Guillermo A, Fox BC, Colomo L, Martinez A, Roncador G et al. High numbers of tumor-infiltrating FOXP3-positive regulatory T cells are associated with improved overall survival in follicular lymphoma. Blood 2006; 108: 2957–2964.
Farinha P, Al-Tourah A, Gill K, Klasa R, Connors JM, Gascoyne RD . The architectural pattern of FOXP3-positive T cells in follicular lymphoma is an independent predictor of survival and histologic transformation. Blood 2010; 115: 289–295.
Day CL, Kaufmann DE, Kiepiela P, Brown JA, Moodley ES, Reddy S et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature 2006; 443: 350–354.
D'Souza M, Fontenot AP, Mack DG, Lozupone C, Dillon S, Meditz A et al. Programmed death 1 expression on HIV-specific CD4+ T cells is driven by viral replication and associated with T cell dysfunction. J Immunol 2007; 179: 1979–1987.
Jin HT, Anderson AC, Tan WG, West EE, Ha SJ, Araki K et al. Cooperation of Tim-3 and PD-1 in CD8 T-cell exhaustion during chronic viral infection. Proc Natl Acad Sci USA 2010; 107: 14733–14738.
Sakuishi K, Apetoh L, Sullivan JM, Blazar BR, Kuchroo VK, Anderson AC . Targeting Tim-3 and PD-1 pathways to reverse T cell exhaustion and restore anti-tumor immunity. J Exp Med 2010; 207: 2187–2194.
Breitfeld D, Ohl L, Kremmer E, Ellwart J, Sallusto F, Lipp M et al. Follicular B helper T cells express CXC chemokine receptor 5, localize to B cell follicles, and support immunoglobulin production. J Exp Med 2000; 192: 1545–1552.
Schaerli P, Willimann K, Lang AB, Lipp M, Loetscher P, Moser B . CXC chemokine receptor 5 expression defines follicular homing T cells with B cell helper function. J Exp Med 2000; 192: 1553–1562.
Kim CH, Rott LS, Clark-Lewis I, Campbell DJ, Wu L, Butcher EC . Subspecialization of CXCR5+ T cells: B helper activity is focused in a germinal center-localized subset of CXCR5+ T cells. J Exp Med 2001; 193: 1373–1381.
Yang ZZ, Grote DM, Ziesmer SC, Niki T, Hirashima M, Novak AJ et al. IL-12 upregulates TIM-3 expression and induces T cell exhaustion in patients with follicular B cell non-Hodgkin lymphoma. J Clin Invest 2012; 122: 1271–1282.
Yang ZZ, Grote DM, Xiu B, Ziesmer SC, Price-Troska TL, Hodge LS et al. TGF-beta upregulates CD70 expression and induces exhaustion of effector memory T cells in B-cell non-Hodgkin's lymphoma. Leukemia 2014; 28: 1872–1878.
Muenst S, Hoeller S, Willi N, Dirnhofera S, Tzankov A . Diagnostic and prognostic utility of PD-1 in B cell lymphomas. Dis Markers 2010; 29: 47–53.
Richendollar BG, Pohlman B, Elson P, Hsi ED . Follicular programmed death 1-positive lymphocytes in the tumor microenvironment are an independent prognostic factor in follicular lymphoma. Hum Pathol 2011; 42: 552–557.
Carreras J, Lopez-Guillermo A, Roncador G, Villamor N, Colomo L, Martinez A et al. High numbers of tumor-infiltrating programmed cell death 1-positive regulatory lymphocytes are associated with improved overall survival in follicular lymphoma. J Clin Oncol 2009; 27: 1470–1476.
Yu D, Rao S, Tsai LM, Lee SK, He Y, Sutcliffe EL et al. The transcriptional repressor Bcl-6 directs T follicular helper cell lineage commitment. Immunity 2009; 31: 457–468.
Bryant VL, Ma CS, Avery DT, Li Y, Good KL, Corcoran LM et al. Cytokine-mediated regulation of human B cell differentiation into Ig-secreting cells: predominant role of IL-21 produced by CXCR5+ T follicular helper cells. J Immunol 2007; 179: 8180–8190.
Trautmann L, Janbazian L, Chomont N, Said EA, Gimmig S, Bessette B et al. Upregulation of PD-1 expression on HIV-specific CD8+ T cells leads to reversible immune dysfunction. Nat Med 2006; 12: 1198–1202.
Myklebust JH, Irish JM, Brody J, Czerwinski DK, Houot R, Kohrt HE et al. High PD-1 expression and suppressed cytokine signaling distinguish T cells infiltrating follicular lymphoma tumors from peripheral T cells. Blood 2013; 121: 1367–1376.
Zhou Q, Munger ME, Veenstra RG, Weigel BJ, Hirashima M, Munn DH et al. Coexpression of Tim-3 and PD-1 identifies a CD8+ T-cell exhaustion phenotype in mice with disseminated acute myelogenous leukemia. Blood 2011; 117: 4501–4510.
Acknowledgements
Supported in part by grants from the National Institutes of Health (P50 CA97274), the Lymphoma Research Foundation, the Leukemia & Lymphoma Society and the Predolin Foundation.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Yang, ZZ., Grote, D., Ziesmer, S. et al. PD-1 expression defines two distinct T-cell sub-populations in follicular lymphoma that differentially impact patient survival. Blood Cancer Journal 5, e281 (2015). https://doi.org/10.1038/bcj.2015.1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bcj.2015.1
- Springer Nature Limited
This article is cited by
-
Characterization of immune exhaustion and suppression in the tumor microenvironment of splenic marginal zone lymphoma
Leukemia (2023)
-
The expression and clinical significance of programmed cell death receptor 1 and its ligand in tumor tissues of patients with extranodal nasal NK/T cell lymphoma
Scientific Reports (2022)
-
Prognostic implication of CTLA-4, PD-1, and PD-L1 expression in aggressive adult T-cell leukemia–lymphoma
Annals of Hematology (2022)
-
Immune infiltrate diversity confers a good prognosis in follicular lymphoma
Cancer Immunology, Immunotherapy (2021)
-
Newly diagnosed follicular lymphoma during pembrolizumab treatment for lung cancer
International Journal of Hematology (2021)