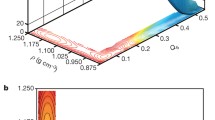
Abstract
MANY of the low-temperature thermodynamic properties of water, such as heat capacity and isothermal compressibility, exhibit anomalous behaviour that tends to diverge in the supercooled state1,2. On the basis of molecular dynamics simulations3,4, these phenomena have recently been attributed to the influence of a critical point terminating the temperature–pressure coexistence line separating low- and high-density amorphous ices. But the fact that water tends to lose its anomalous behaviour5 in the pressure range predicted for the new critical point poses problems for such an interpretation. Moreover, the phase diagram derived from these simulations contrasts sharply with another conjecture, whereby it is argued that at atmospheric pressure no thermodynamically continuous path exists between low-density amorphous ice and normal water6. Here I report the results of a series of molecular dynamics simulations at constant (approximately atmospheric) pressure, which show that both ideas can be reconciled by relocating the critical point to negative pressures. The resulting phase diagram is not only simpler, but also accounts for the transition of water from a fragile to a strong liquid in the supercoooled region7,8.
Similar content being viewed by others
References
Speedy, R. J. & Angell, C. A. J. chem. Phys. 65, 851–858 (1976).
Angell, C. A., Oguni, M. & Sichina, W. J. J. phys. Chem. 86, 998–1002 (1982).
Poole, P. H., Sciortino, F., Essmann, U. & Stanley, H. E. Nature 360, 324–328 (1992).
Poole, P. H., Sciortino, F., Essmann, U. & Stanley, H. E. Phys. Rev. E48, 3799–3817 (1993).
Eisenberg, D. & Kauzmann, W. The Structure and Properties of Water (Oxford Univ. Press, London, 1969).
Speedy, R. J. J. phys. Chem. 96, 2322–2325 (1992).
Angell, C. A. J. phys. Chem. 97, 6339–6341 (1993).
Angell, C. A. in Hydrogen Bond Networks (eds Bellissent-Funel, M-. C. & Dore, J. C.) 3–22 (Kluwer, Dortrecht, 1994).
Nosé, S. Molec. Phys. 52, 255–268 (1984).
Nosé, S. J. chem. Phys. 81, 511–519 (1984).
Andersen, H. C. J. chem. Phys. 72, 2384–2393 (1980).
Jorgensen, W. L., Chandrasekhar, J., Madura, J. D., Impey, R. W. & Klein, M. L. J. chem. Phys. 79, 926–935 (1983).
Poole, P. H., Essmann, U., Sciortino, F. & Stanley, H. E. Phys. Rev. E48, 4605–4610 (1993).
Stillinger, F. H. & Weber, T. A. J. phys. Chem. 87, 2833–2840 (1983).
Stillinger, F. H. J. chem. Phys. 88, 7818–7825 (1988).
Hallbrucker, A. & Mayer, E. J. phys. Chem. 91, 503–505 (1987).
Stanley, H. E. et al. Physica A205, 122–139 (1994).
Ponyatovskii, E. G., Sinitsyn, V. V. & Pozdnyakova, T. A. JETP Lett. 60, 360–364 (1994).
Bellissent-Funel, M.-C. & Bosio, L. J. chem. Phys. 102, 3727–3735 (1995).
Poole, P. H., Sciortino, F., Grande, T., Stanley, H. E. & Angell, C. A. Phys. Rev. Lett. 73, 1632–1635 (1994).
Mishima, O., Colvert, L. D. & Whalley, E. Nature 310, 393–394 (1984).
Mishima, O., Colvert, L. D. & Whalley, E. Nature 314, 76–78 (1985).
Speedy, R. J. J. phys. Chem. 86, 982–991 (1982).
Sceats, M. S. & Rice, S. A. J. chem. Phys. 72, 3260–3262 (1980).
Ohmine, I. & Tanaka, H. J. chem. Phys. 93, 8138–8147 (1990).
Kauzmann, W. Chem. Rev. 43, 219–256 (1948).
Stanley, H. E. & Teixeira, J. J. chem. Phys. 73, 3404–3422 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tanaka, H. A self-consistent phase diagram for supercooled water. Nature 380, 328–330 (1996). https://doi.org/10.1038/380328a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/380328a0
- Springer Nature Limited
This article is cited by
-
Inter-enantiomer conversion dynamics and Johari–Goldstein relaxation of benzophenones
Scientific Reports (2021)
-
Critical behavior of a water monolayer under hydrophobic confinement
Scientific Reports (2014)
-
Water at Biological and Inorganic Interfaces
Food Biophysics (2013)
-
Interplay of the Glass Transition and the Liquid-Liquid Phase Transition in Water
Scientific Reports (2012)
-
Understanding the role of hydrogen bonds in water dynamics and protein stability
Journal of Biological Physics (2012)