Abstract
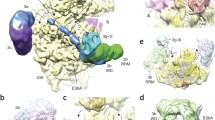
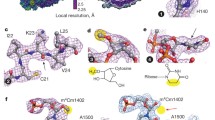
The crystal structure of the EF-Tu·EF-Ts complex from Escherichia coli has been determined to a resolution of 2.5 Å. The complex contains two subunits of each of the elongation factors. The two EF-Ts molecules form a tight dirtier, but there is little contact between the two EF-Tu molecules. The interaction of EF-Ts with EF-Tu results principally in the disruption of the Mg2+ ion binding site, thereby reducing the affinity of EF-Tu for guanine nucleotides.
Similar content being viewed by others
References
Ertel, R., Redfield, B., Brot, N. & Weissbach, H. Archs Biochem. Biophys. 128, 331–338 (1968).
Bourne, H. R., Sanders, D. A. & McCormick, F. Nature 349, 117–127 (1991).
Lucus-Lenard, J. & Lipmann, F. Proc. natn. Acad. Sci. U.S.A. 55, 1562–1566 (1966).
Weissbach, H., Miller, D. L. & Hachmann, J. Archs Biochem. Biophys. 137, 262–269 (1970).
Miller, D. L. & Weissbach, H. Archs Biochem. Biophys. 141, 26–37 (1970).
Leberman, R. et al. Analyt. Biochem. 104, 29–36 (1980).
Blumental, T., Landers, T. A. & Weber, K. Proc. natn. Acad. Sci. U.S.A. 69, 1313–1317 (1972).
Kjeldgaard, M. & Nyborg, J. J. molec. Biol. 223, 721–742 (1992).
Kabsch, W., Gast, W. H., Schulz, G. E. & Leberman, R. J. molec. Biol. 117, 999–1012 (1977).
Pai, E. F. et al. Nature 341, 209–214 (1989).
Kjeldgaard, M., Nissen, P., Thirup, S. & Nyborg, J. Structure 1, 35–50 (1993).
Berchtold, H. et al. Nature 365, 126–132 (1993).
Jones, M. D. et al. Eur. J. Biochem. 108, 507–526 (1980).
An, G., Bendiak, D. S., Mamelak, L. A. & Friesen, J. D. Nucleic Acids Res. 9, 4163–4172 (1981).
Leberman, R., Schulz, G. E. & Suck, D. FEBS Lett. 124, 279–281 (1981).
Schneider, U. dissertation, Univ. Freiburg, Germany (1983).
Schick, B. & Jurnak, F. Acta crystallogr. D50, 563–568 (1994).
Kawashima, T., Berthet-Colominas, C., Cusask, S. & Leberman, R. Acta crystallogr. (in the press).
Collaborative Computing Project No. 4 (SERC Daresbury Laboratory, Warrington, 1979).
Arai, K.-I., Kawakita, M. & Kaziro, Y. J. biol Chem. 247, 7029–7037 (1972).
John, J. et al. J. biol. Chem. 268, 923–929 (1993).
Blumental, T. & Carmichael, G. C. A. Rev. Biochem. 48, 525–548 (1979).
Janssen, G. M. C., van Damme, H. T. F., Kriek, J., Amons, R. & Möller, W. J. biol. Chem. 269, 32410–32417 (1994).
Boguski, M. S. & McCormick, G. Nature 366, 643–654 (1993).
Leslie, A. G. W. in Proceedings of the CCP4 Study Weekend (eds Helliwell, J. R., Machin, P. A. & Papiz, M. Z.) 39–50 (SERC Daresbury Laboratory, Warrington, 1987).
Jones, T. A., Cowan, S., Zou, J.-Y. & Kjelgaard, M. Acta crystallogr. A47, 110–119 (1990).
Jones, T. A. in Molecular Replacement (eds Dodson, E. J., Gover, S. & Wolf, W.) 92–105 (SERC Daresbury Laboratory, Warrington, 1992).
Brünger, A. T. Nature 355, 472–475 (1992).
Kraulis, P. J. appl. Crystallogr. 24, 946–950 (1991).
Kabsh, W. & Sander, C. Biopolymers 22, 2577–2637 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kawashima, T., Berthet-Colominas, C., Wulff, M. et al. The structure of the Escherichia coli EF-Tu· EF-Ts complex at 2.5 Å resolution. Nature 379, 511–518 (1996). https://doi.org/10.1038/379511a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/379511a0
- Springer Nature Limited
This article is cited by
-
Structural insights of the elongation factor EF-Tu complexes in protein translation of Mycobacterium tuberculosis
Communications Biology (2022)
-
A conserved P-loop anchor limits the structural dynamics that mediate nucleotide dissociation in EF-Tu
Scientific Reports (2015)
-
Genome-wide analysis of alternative splicing in Volvox carteri
BMC Genomics (2014)
-
Interchanging Functionality Among Homologous Elongation Factors Using Signatures of Heterotachy
Journal of Molecular Evolution (2013)
-
π–π Interactions in Structural Stability: Role in RNA Binding Proteins
Cell Biochemistry and Biophysics (2013)