Abstract
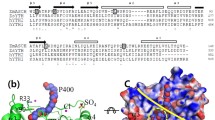
TRANSCRIPTION elongation factors stimulate the activity of DNA-dependent RNA polymerases by increasing the overall elongation rate and the completion of RNA chains. One group of such factors, which includes Escherichia coli GreA, GreB and eukaryotic SII (TFIIS), acts by inducing hydrolytic cleavage of the transcript within the RNA polymerase, followed by relase of the 3′-terminal fragment1–5. Here we report the crystal structure of GreA at 2.2 Å resolution. The structure contains an amino-terminal domain consisting of an antiparallel α-helical coiled-coil dimer which extends into solution, reminiscent of the coiled coil in seryl-tRNA synthetases6. A site near the tip of the coiled-coil "finger" plays a direct role in the transcript cleavage reaction by contacting the 3'-end of the transcript. The structure exhibits an unusual asymmetric charge distribution which indicates the manner in which GreA interacts with the RNA polymerase elongation complex.
Similar content being viewed by others
References
Izban, M. G. & Luse, D. S. Genes Dev. 6, 1342–1356 (1992).
Reines, D. J. biol. Chem. 267, 3795–3800 (1992).
Borukhov, S., Polyakov, A., Nikiforov, V. & Goldfarb, A. Proc. natn. Acad. Sci. U.S.A. 89, 8899–8902 (1992).
Borukhov, S., Sagitov, V. & Goldfarb, A. Cell 72, 459–466 (1993).
Kassavetis, G. A. & Geiduschek, E. P. Science 259, 944–945 (1993).
Cusack, S., Berthet-Colominas, C., Härtlein, M., Nassar, N. & Leberman, R. Nature 347, 249–255 (1990).
Harrison, C. J., Bohm, A. A. & Nelson, H. C. M. Science 263, 224–227 (1994).
Woody, A. Y., Vader, C. R., Woody, R. W. & Haley, B. E. Biochemistry 23, 2843–2848 (1984).
Borukhov, S., Lee, J. & Goldfarb, A. J. biol. Chem. 266, 23932–23935 (1991).
Marks, G. L & Wood, D. O. Nucleic Acids Res. 20, 3785 (1992).
Biou, V., Yaremchuk, A., Tukalo, M. & Cusack, S. Science 263, 1404–1410 (1994).
Darst, S. A. et al. J. molec. Biol. 242, 582–585 (1994).
Hendrickson, W., Norton, J. R. & LeMaster, D. M. EMBO J. 9, 1665–1672 (1990).
Zhang, K. Y. J. & Main, P. Acta crystallogr. A46, 41–46 (1990).
Jones, T. A., Zou, J.-Y., Cowan, S. & Kjeldgaard, M. Acta crystallogr. A47, 110–119 (1991).
Brünger, A. X-PLOR (Version 3.1) Manual (Yale University, Connecticut, 1992).
Read, R. Acta crystallogr. A42, 140–149 (1986).
Ramachandran, G. N., Ramakrishnan, C. & Sasisekharan, V. J. molec. biol. 7, 95–99 (1963).
Bernstein, F. C. et al. J. molec. Biol. 112, 535–542 (1977).
Richardson, J. M. Meth. Enzym. 115, 359–390 (1985).
Kabsch, W. & Sander, C. Biopolymers 22, 2577–2637 (1983).
Nicholls, A., Sharp, K. A. & Honig, B. Prot. Struct. Funct. Genet. 11, 281–296 (1991).
Sparkowski, J. & Das, A. Nucleic Acids Res. 18, 6443 (1990).
Jacobson, G. R., Schaffer, M. H., Stark, G. R. & Vanaman, T. C. J. biol. Chem. 248, 6583–6591 (1973).
Schagger, H. & von Jagow, G. Analyt. Biochem. 166, 368–379 (1987).
Drapeau, G. R. J. biol. Chem. 255, 839–840 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Stebbins, C., Borukhov, S., Orlova, M. et al. Crystal structure of the GreA transcript cleavage factor from Escherichia coli. Nature 373, 636–640 (1995). https://doi.org/10.1038/373636a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/373636a0
- Springer Nature Limited
This article is cited by
-
Mutational analysis of Escherichia coli GreA protein reveals new functional activity independent of antipause and lethal when overexpressed
Scientific Reports (2020)
-
Replication–transcription conflicts in bacteria
Nature Reviews Microbiology (2012)
-
Crystal structure of bacterial RNA polymerase bound with a transcription inhibitor protein
Nature (2010)
-
Super DksAs: substitutions in DksA enhancing its effects on transcription initiation
The EMBO Journal (2009)
-
The carboxy‐terminal coiled‐coil of the RNA polymerase β′‐subunit is the main binding site for Gre factors
EMBO reports (2007)