Abstract
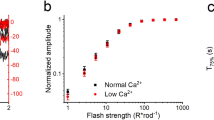
DURING light-adaptation by the vertebrate eye, the rods are desensitized and the light response is accelerated1,2. When light is absorbed by the rods, a phosphodiesterase is activated that hydrolyses cyclic GMP3,4. A light-induced decrease in cytoplasmic Ca2+ concentration5–7 is part of this light-adaptation process8,9. The protein S-modulin (Mr 26,000) is known to increase the fraction of light-activated cyclic GMP-phosphodiesterase (PDE) at high Ca2+ concentrations in frog rod photoreceptors10. Here I present evidence that S-modulin lengthens the lifetime of active PDE (PDE*) at high Ca2+ concentrations. These S-modulin effects are observed in the physiological range of Ca2+ concentration (30 nM to 1 µM; half-maximum effects at 200–400 nM). At the high Ca2+ concentrations at which S-modulin prolongs the lifetime of PDE*, S-modulin inhibits rhodopsin phosphorylation (half-maximum effect at ˜100 nM Ca2+). ATP is necessary for the S-modulin effects on PDE activation. I therefore conclude that the Ca2+-dependent regulation of PDE by S-modulin is mediated by rhodopsin phosphorylation. This regulation seems to be the principal mechanism of light adaptation in vertebrate photoreceptors.
Similar content being viewed by others
References
Baylor, D. A. & Hodgkin, A. L. J. Physiol. 242, 729–758 (1974).
Baylor, D. A., Lamb, T. D. & Yau, K.-W. J. Physiol. 288, 589–611 (1979).
Stryer, L. A. Rev. Neurosci. 9, 87–119 (1986).
Kaupp, U. B. & Koch, K.-W. A. Rev. Physiol. 54, 153–175 (1992).
Yau, K.-W. & Nakatani, K. Nature 313, 579–582 (1985).
McNaughton, P. A., Cervetto, L. & Nunn, B. J. Nature 322, 261–263 (1986).
Ratto, G. M., Payne, R., Owen, W. G. & Tsien, R. Y. J. Neurosci. 8, 3240–3246 (1988).
Matthews, H. R., Murphy, R. L. W., Fain, G. & Lam, T. D. Nature 334, 67–69 (1988).
Nakatani, K. & Yau, K.-W. Nature 334, 69–71 (1988).
Kawamura, S. & Murakami, M. Nature 349, 420–423 (1991).
Kawamura, S., Takamatsu, K. & Kitamura, K. Biochem. biophys. Res. Commun. 186, 411–417 (1992).
Kawamura, S. & Bownds, M. D. J. gen. Physiol. 77, 571–591 (1981).
Barkdoll, A. E. III, Pugh, E. N. Jr & Sitaramayya, A. J. gen. Physiol. 93, 1091–1108 (1989).
Torre, V., Matthews, H. R. & Lamb, T. D. Proc. natn. Acad. Sci. U.S.A. 83, 7109–7113 (1986).
Liebman, P. A. & Pugh, E. N. Jr Nature 287, 734–736 (1980).
Kawamura, S. Biochim. biophys. Acta 732, 276–281 (1983).
Dizhoor, A. M. et al. Science 251, 915–918 (1991).
Lambrecht, H.-G. & Koch, K.-W. EMBO J. 10, 793–798 (1991).
Benovic, J. L., Bouvier, M., Caron, M. G. & Lefkowitz, R. J. A. Rev. Cell Biol. 4, 405–428 (1988).
Kawamura, S. & Murakami, M. Biochim. biophys. Acta 870, 256–266 (1986).
Yee, R. & Liebman, P. A. J. biol. Chem. 253, 8902–8909 (1978).
Kawamura, S. & Murakami, M. J. gen. Physiol. 94, 649–668 (1989).
Laemmli, U. K. Nature 227, 680–685 (1970).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kawamura, S. Rhodopsin phosphorylation as a mechanism of cyclic GMP phosphodiesterase regulation by S-modulin. Nature 362, 855–857 (1993). https://doi.org/10.1038/362855a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/362855a0
- Springer Nature Limited
We’re sorry, something doesn't seem to be working properly.
Please try refreshing the page. If that doesn't work, please contact support so we can address the problem.