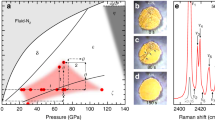
Abstract
WEAKLY bound van der Waals molecules, consisting of atoms or molecules (such as rare gases, H2 and N2) interacting through van der Waals forces, can be formed in the gas phase1. The existence of similar weakly bound stoichiometric compounds in condensed phases has remained an open question as the components usually form separate pure phases or solid solutions2. Here we report on a detailed study of the helium–nitrogen system in a diamond-anvil cell using synchrotron X-ray diffraction, Raman scattering and optical microscopy. We find that high pressure stabilizes the formation of a stoichiometric, solid van der Waals compound of composition He(N2)11. Because of the relatively simple interactions involved, this solid may exemplify a novel class of van der Waals compounds that are formed only at high pressures. Van der Waals chemistry in simple molecular systems may be important for understanding the structure and properties of the interiors of the outer planets and their satellites, where pressures are high and such components may be abundant3–5.
Similar content being viewed by others
References
Novick, S. E., Leopold, K. R. & Klemperer, W. in Atomic and Molecular Clusters (ed. Bernstein, E. R.) 359–391 (Elsevier, New York, 1990).
Meyer, L. Adv. chem. Phys. 16, 343–387 (1969).
Hubbard, W. B. Planetary Interiors (van Nostrand, New York, 1984).
Nellis, W. J. et al. Science 240, 779–781 (1988).
Hubbard, W. B. et al. Science 253, 648–651 (1991).
Schouten, J. A. Phys. Rep. 172, 33–92 (1989).
Schouten, J. A. Fluid Phase Equil. (in the press).
Cromer, D. T., Mills, R. L., Schiferl, D. & Schwalbe, L. A. Acta crystallogr. B37, 8–11 (1981).
Mills, R. L., Cromer, D. T. & Olinger, B. J. chem. Phys. 84, 2837–2845 (1986).
Olijnyk, H. J. chem. Phys 93, 8968–8972 (1990).
Vos, W. L., van Hinsberg, M. G. E. & Schouten, J. A. Phys. Rev. B42, 6106–6109 (1990).
Mao, H. K. et al. Phys. Rev. Lett. 60, 2649–2652 (1988).
Vos, W. L. & Schouten, J. A. Phys. Rev. Lett. 64, 898–901 (1990).
Scheerboom, M. I. M. & Schouten, J. A. J. Phys.: Condensed Matter 3, 8305–8312 (1991).
Vos, W. L. & Schouten, J. A. Physica A182, 365–387 (1992).
van den Bergh, L. C., Schouten, J. A. & Trappeniers, N. J. Physica A141, 524–538 (1987).
Mao, H. K. et al. Science 239, 1131–1134 (1988).
Le Toullec, R., Loubeyre, P. & Pinceaux, J.-P. Phys. Rev. B40, 2368–2378 (1990).
van den Bergh, L. C. & Schouten, J. A. J. chem. Phys. 89, 2336–2343 (1988).
Bartlett P. J. Phys.: Condensed Matter 2, 4979–4989 (1990).
Murray, M. J. & Sanders, J. V. Phil. Mag. A42, 721–740 (1980).
Kitaigorodsky, A. I. Mixed Crystals (Springer, Berlin, 1984).
Hemley, R. J. & Mao, H. K. in Proc. APS 1991 Topical Conf. Shock Compression of Condensed Matter (eds Schmidt, S. C., Dick, R. D., Forbes, J. W. & Tasker, D. G.) (in the press).
Mao, H. K. in Simple Molecular Systems at Very High Densities (eds Polian, A., Loubeyre, P. & Boccara, N.) 221–236 (Plenum, New York, 1989).
Hu, J. Z., Mao, H. K. & Shu, J. F. EOS 72, 554 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vos, W., Finger, L., Hemley, R. et al. A high-pressure van der Waals compound in solid nitrogen-helium mixtures. Nature 358, 46–48 (1992). https://doi.org/10.1038/358046a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/358046a0
- Springer Nature Limited
This article is cited by
-
Creating two-dimensional solid helium via diamond lattice confinement
Nature Communications (2022)
-
High pressure synthesis of phosphine from the elements and the discovery of the missing (PH3)2H2 tile
Nature Communications (2020)
-
Multiple superionic states in helium–water compounds
Nature Physics (2019)
-
Electrostatic force driven helium insertion into ammonia and water crystals under pressure
Communications Chemistry (2019)
-
Route to high-energy density polymeric nitrogen t-N via He−N compounds
Nature Communications (2018)