Abstract
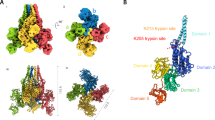
The structure of the δ-endotoxin from Bacillus thuringiensis subsp. tenebrionis that is specifically toxic to Coleopterajnsects (beetle toxin) has been determined at 2.5 Å resolution. It comprises three domains which are, from the N- to C-termini, a seven-helix bundle, a three-sheet domain, and a β sandwich. The core of the molecule encompassing all the domain interfaces is built from conserved sequence segments of the active δ-endotoxins. Therefore the structure represents the general fold of this family of insecticidal proteins. The bundle of long, hydrophobic and amphipathic helices is equipped for pore formation in the insect membrane, and regions of the three-sheet domain are probably responsible for receptor binding.
Similar content being viewed by others
References
Höfte, H. & Whiteley, H. R., Microbiol. Rev. 53, 242–255 (1989).
Ellar, D. J. et al. in Molecular Biology of Microbial Differentiation (eds Hoch, J. A. & Setlow, P.) 230–240 (Am. Soc. Microbiol., Washington, DC, 1985).
Wilcox, E. R. et al. in Protein Engineering: Applications in Science, Medicine and Industry (eds Inouye, M. & Sarma, R.) 395–413 (Academic. New York, 1986).
Vaeck, M. et al. Nature 328, 33–37 (1987).
Perlak, F. J., Fuchs, R. L., Dean, D. A., McPherson, D. L. & Fischhoff, D. A. Proc. natn. Acad. Sci. U.S.A. 88, 3324–3328 (1991).
Barton, K. A., Whiteley, H. R. & Yang, N. S. Plant Physiol. 85, 1103–1109 (1987).
Hofmann, C., Lüthy, P., Hütter, R. & Pliska, V. Eur. J. Biochem. 173, 85–91 (1988).
Hofmann, C. et al. Proc. natn. Acad. Sci. U.S.A. 85, 7844–7848 (1988).
Van Rie, J., Jansens, S., Höfte, H., Degheele, D. & Van Mellaert, H. Appl. Envir. Microbiol. 56, 1378–1385 (1990).
Knowles, B. H. & Ellar, D. J. Biochim. biophys. Acta 924, 509–518 (1987).
Endo, Y. & Nishitsutsuji-Uwo, J. J. Invertebr. Path. 36, 90–103 (1980).
Hodgman, T. C. & Ellar, D. J. J. DNA Sequ. Map. 1, 97–106 (1990).
Ge, A. Z., Shivarova, N. I. & Dean, D. H. Proc. natn. Acad. Sci. U.S.A. 86, 4037–4041 (1989).
Widner, W. R. & Whiteley, H. R. J. Bact. 172, 2826–2832 (1990).
Schnepf, H. E., Tomczak, K., Ortega, J. P. & Whiteley, H. R. J. biol. Chem. 265, 20923–20939 (1990).
Krieg, A., Huger, A. M., Langenbruch, G. A. & Schnetter, W. J. appl. Entomol. 96, 500–508 (1983).
Höfte, H., Seurinck, J., Van Houtven, A. & Vaeck, M. Nucleic Acids Res. 15, 7183 (1987).
Li, J., Henderson, R., Carroll, J. & Ellar, D. J. molec. Biol. 199, 543–545 (1988).
Carroll, J., Li, J. & Ellar, D. J. Biochem. J. 261, 99–105 (1989).
Arndt, U. W. Meth. Enzym. 114, 472–485 (1985).
Wang, B. C. Meth. Enzym. 115, 90–112 (1985).
Leslie, A. G. W. Acta crystallogr. A43, 134–136 (1987).
Jones, T. A., Zou, J.-Y., Cowan, S. W. & Kjeldgaard, N. Acta crystallogr. A47, 110–119 (1991).
Brünger, A. T. J. molec. Biol. 203, 803–816 (1988).
Chothia, C. & Finkelstein, A. V. A. Rev. Biochem. 59, 1007–1039 (1990).
Chothia, C. J. molec. Biol 105, 1–15 (1976).
Burley, S. K. & Petsko, G. A. Science 229, 23–28 (1985).
Parker, M. W., Pattus, F., Tucker, A. D. & Tsernoglou, D. Nature 337, 93–96 (1989).
Richardson, J. S. Adv. Prot. Chem. 34, 167–339 (1981).
Höfte, H. et al. Eur. J. Biochem 161, 273–280 (1986).
Choma, C. T. & Kaplan, H. Biochemistry 29, 10971–10977 (1990).
Nicholls, C. N., Ahmad, W. & Ellar, D. J. J. Bact. 171, 5141–5147 (1989).
MacIntosh, S. C., McPherson, S. L., Perlak, F. J., Marrone, P. G. & Fuchs, R. L. Biochem. biophys. Res. Commun. 170, 665–672 (1990).
Schnepf, H. E. & Whiteley, H. R. J. biol. Chem. 260, 6273–6280 (1985).
Adang, M. J. et al. Gene 36, 289–300 (1985).
Sekar, R., Thompson, D. V., Maroney, M. J., Bookland, R. G. & Adang, M. J. Proc. natn. Acad. Sci. U.S.A. 84, 7036–7040 (1987).
Chungjatupornchai, W., Höfte, H. Seurinck, J., Angsuthanasombat, C. & Vaeck, M. Eur. J. Biochem. 173, 9–16 (1988).
Knowles, B. R. Thomas, W. E. & Ellar, D. J. FEBS Lett. 168, 197–202 (1984).
Knowles, B. H. & Ellar, D. J. J. Cell Sci. 83, 89–101 (1986).
Haider, M. Z. & Ellar, D. J. Biochem. J. 248, 197–201 (1987).
Visser, B., Munsterman, E., Stoker, A. & Dirske, W. G. J. Bact. 172, 6783–6788 (1990).
Lakey, J. H., Baty, D. & Pattus, F. J. molec. Biol. 218, 639–653 (1991).
Song, H. Y., Cohen, F. S. & Cramer, W. A. J. Bact. 173, 2927–2934 (1991).
Ahmad, W. & Ellar, D. J. FEMS Microbiol. Lett. 68, 97–104 (1990).
Messerschimidt, A. & Pflugrath, J. W. J. appl. Crystallogr. 20, 306–315 (1987).
Read, R. J. Acta crystallogr. A42, 140–149 (1986).
Kraulis, P. J. appl. Crystallogr. (in the press).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Li, J., Carroll, J. & Ellar, D. Crystal structure of insecticidal δ-endotoxin from Bacillus thuringiensis at 2.5 Å resolution. Nature 353, 815–821 (1991). https://doi.org/10.1038/353815a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/353815a0
- Springer Nature Limited
This article is cited by
-
Whole genome sequencing of a novel Bacillus thuringiensis isolated from Assam soil
BMC Microbiology (2023)
-
Structural, functional, and evolutionary analysis of Cry toxins of Bacillus thuringiensis: an in silico study
Egyptian Journal of Biological Pest Control (2021)
-
Real-time monitoring of bacterial growth kinetics in suspensions using laser speckle imaging
Scientific Reports (2020)
-
Directed evolution of a genetically encoded immobilized lipase for the efficient production of biodiesel from waste cooking oil
Biotechnology for Biofuels (2019)
-
The crystal structure of the chitinase ChiA74 of Bacillus thuringiensis has a multidomain assembly
Scientific Reports (2019)