Abstract
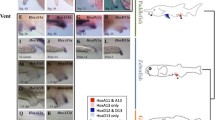
Genes of the Hox cluster are restricted to the animal kingdom and play a central role in axial patterning in divergent animal phyla1. Despite its evolutionary and developmental significance, the origin of the Hox gene cluster is obscure. The consensus is that a primordial Hox cluster arose by tandem gene duplication close to animal origins2,3,4,5. Several homeobox genes with high sequence identity to Hox genes are found outside the Hox cluster and are known as ‘dispersed’ Hox-like genes; these genes may have been transposed away from an expanding cluster6. Here we show that three of these dispersed homeobox genes form a novel gene cluster in the cephalochordate amphioxus. We argue that this ‘ParaHox’ gene cluster is an ancient paralogue (evolutionary sister) of the Hox gene cluster; the two gene clusters arose by duplication of a ProtoHox gene cluster. Furthermore, we show that amphioxus ParaHox genes have co-linear developmental expression patterns in anterior, middle and posterior tissues. We propose that the origin of distinct Hox and ParaHox genes by gene-cluster duplication facilitated an increase in body complexity during the Cambrian explosion.





Similar content being viewed by others
References
Slack, J. M. W., Holland, P. W. H. & Graham, C. F. The zootype and the phylotypic stage. Nature 361, 490–492 (1993).
Kappen, C., Schughart, K. & Ruddle, F. H. Two steps in the evolution of Antennapedia-class vertebrate homeobox genes. Proc. Natl Acad. Sci. USA 86, 5458–5463 (1989).
Schubert, F. R., Nieselt-Struwe, K. & Gruss, P. The Antennapedia-type homeobox genes have evolved from three precursors separated early in metazoan evolution. Proc. Natl Acad. Sci. USA 90, 143–147 (1993).
Gehring, W. J., Affolter, M. & Bürglin, T. Homeodomain proteins. Annu. Rev. Biochem. 63, 487–526 (1994).
Zhang, J. & Nei, M. Evolution of Antennapedia-class homeobox genes. Genetics 142, 295–303 (1996).
Bürglin, T. in A Guidebook for Homeobox Genes (ed. Duboule, D.) 25–71 (Oxford University Press, Oxford, 1994).
Miller, D. J. & Miles, A. Homeobox genes and the zootype. Nature 365, 215–216 (1993).
Candia, A. F. et al. Mox-1 and Mox-2 define a novel homeobox gene subfamily and are differentially expressed during early mesodermal patterning in mouse embryos. Development 116, 1123–1136 (1992).
Garcia-Fernàndez, J. & Holland, P. W. H. Archetypal organization of the amphioxus Hox gene cluster. Nature 370, 563–566 (1994).
Averof, M., Dawes, R. & Ferrier, D. Diversification of arthropod Hox genes as a paradigm for the evolution of gene functions. Sem. Cell. Dev. Biol. 7, 539–551 (1996).
Williams, N. A. & Holland, P. W. H. Old head on young shoulders. Nature 383, 490 (1996).
Duprey, P. et al. Amouse gene homologous to the Drosophila gene caudal is expressed in epithelial cells from the embryonic intestine. Genes Dev. 2, 1647–1654 (1988).
Calleja, M., Moreno, E., Pelaz, S. & Morata, G. Visualization of gene expression in living adult Drosophila. Science 274, 252–255 (1996).
Wright, C. V. E., Schnegelsberg, P. & DeRobertis, E. M. XIHbox 8—a novel Xenopus homeoprotein restricted to a narrow-band of endoderm. Development 104, 787–794 (1988).
Offield, M. F. et al. Pdx-1 is required for pancreatic outgrowth and differentiation of the rostral duodenum. Development 122, 983–995 (1996).
Wysocka-Diller, J., Aisemberg, G. O. & Macagmo, E. R. Anovel homeobox cluster expressed in repeated structures of the midgut. Dev. Biol. 171, 439–447 (1995).
Jonsson, J., Carlsson, L., Edlund, T. & Edlund, H. Insulin-promoter-factor-1 is required for pancreas development in mice. Nature 371, 606–609 (1994).
Szucsik, J. C. et al. Altered forebrain and hindbrain development in mice mutant for the Gsh-2 homeobox gene. Dev. Biol. 191, 230–242 (1997).
Li, H., Zeitler, P. S., Valerius, M. T., Small, K. & Potter, S. S. Gsh-1, an orphan Hox gene, is required for normal pituitary development. EMBO J. 15, 714–724 (1996).
Gorbman, A. Early development of the hagfish pituitary gland: evidence for the endodermal origin of the adenohypophysis. Am. Zool. 23, 639–654 (1983).
Gaunt, S. J., Sharpe, P. T. & Duboule, D. Spatially restricted domains of homeogene transcripts in mouse embryos: relation to a segmented body plan. Development (suppl.) 104, 169–180 (1988).
Popodi, E., Kissinger, J. C., Andrews, M. E. & Raff, R. A. Sea urchin Hox genes: insights into the ancestral Hox cluster. Mol. Biol. Evol. 13, 1078–1086 (1996).
Irvine, S. Q., Warinner, S. A., Hunter, J. D. & Martindale, M. Q. Asurvey of homeobox genes in Chaetopterus variopedatus and analysis of polychaete homeodomains. Mol. Phylo. Evol. 7, 331–345 (1997).
Stoffel, M. et al. Localization of human homeodomain transcription factor insulin promoter factor-1 (Ipf1) to chromosome band 13q12.1. Genomics 28, 125–126 (1995).
Kozak, C. A., Goffinet, A. & Stephenson, D. A. Mouse chromosome-5. Mamm. Genome 5, s65–s78 (1994).
Fiedorek, F. T. & Kay, E. S. Mapping of the insulin promoter factor-1 gene (Ipf1) to distal mouse chromosome-5. Genomics 28, 581–584 (1995).
Conway Morris S. Why molecular biology needs palaeontology. Development (suppl.) 1–13 (1994).
Lehrach, H. et al. in Genome Analysis 1: Genetic and Physical Mapping (eds Davies, K. E. & Tilghman, S.M.) 38–81 (Cold Spring Harbor Laboratory Press, New York, 1990).
Felsenstein, J. PHYLIP version 3.5c(Department of Genetics, Univ. Washington, Seattle, 1993).
Holland, P. W. H., Holland, L. Z., Williams, N. A. & Holland, N. D. An amphioxus homeobox gene: sequence conservation, spatial expression during development and insights into vertebrate evolution. Development 116, 653–661 (1992).
Acknowledgements
We thank G. Balavoine and C. V. E. Wright for discussion, S. J. Patton and C. Burgtorf for access to the cosmid library and help with screening, and N. A. Williams and S. A. J. Thompson for advice and assistance. This research was funded by a BBSRC Earmarked Studentship (to N.M.B.) and by the DGICYT (J.G.F.), and facilitated by a grant from the Acciones Integradas of the British Council/Ministerio de Educación y Ciencia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brooke, N., Garcia-Fernàndez, J. & Holland, P. The ParaHox gene cluster is an evolutionary sister of the Hox gene cluster. Nature 392, 920–922 (1998). https://doi.org/10.1038/31933
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/31933
- Springer Nature Limited
This article is cited by
-
Sea cucumbers: an emerging system in evo-devo
EvoDevo (2024)
-
Repetitive and zonal expression profiles of absorption-related genes in the gastrointestinal tract of ascidian Ciona intestinalis type A
Cell and Tissue Research (2023)
-
Non-collinear Hox gene expression in bivalves and the evolution of morphological novelties in mollusks
Scientific Reports (2021)
-
Evidence from oyster suggests an ancient role for Pdx in regulating insulin gene expression in animals
Nature Communications (2021)
-
Horseshoe crab genomes reveal the evolution of genes and microRNAs after three rounds of whole genome duplication
Communications Biology (2021)