Abstract
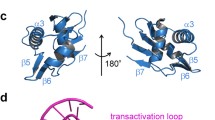
The crystal structure of the Escherichia coli trp repressor has been solved to atomic resolution. The dimeric protein has a remarkable subunit interface in which five of each subunit's six helices are interlinked. The binding of L-tryptophan activates the aporepressor indirectly by fixing the orientation of the second helix of the helix–turn–helix motif and by moulding the details of the repressor's structure near the DNA binding surface.
Similar content being viewed by others
References
Zurawski, G., Gunsalus, R. P., Brown, K. D. & Yanofsky, C. J. molec. Biol. 145, 47–73 (1981).
Kelley, R. L. & Yankofsky, C. Proc. natn. Acad. Sci. U.S.A. 79, 3120–3124 (1982).
Joachimiak, A. J., Kelley, R. L., Gunsalus, R. P., Yanofsky, C. & Sigler, P. B. Proc. natn. Acad. Sci. U.S.A. 80, 668–672 (1983).
Haydock, P. V. & Somerville, R. L. Biochem. biophys. Res. Commun. 119, 926–932 (1984).
Joachimiak, A., Schevitz, R. W., Kelley, R. L., Yanofsky, C. & Sigler, P. B. J. biol. Chem. 258, 12641–12643 (1983).
Anderson, W. F., Ohlendorf, D. H., Takeda, Y. & Matthews, B. W. Nature 290, 754–758 (1981).
Pabo, C. O. & Lewis, M. Nature 298, 443–497 (1982).
McKay, D. B. & Steitz, T. A. Nature 290, 744 (1981).
Tsapakos, M. J., Haydock, P. V., Hermodson, M. & Somerville, R. L. J. biol. Chem. (in the press).
Kelley, R. L. & Yanofsky, C. Proc. natn. Acad. Sci. U.S.A. 82, 483–487 (1985).
Hendrickson, W. A. & Konnert, J. H. in Biomolecular Structure, Conformation and Evolution Vol. 1 (ed. Srinivassan, R.) 43–57 (Pergamon, Oxford, 1980).
Jones, T. A. in Computational Crystallography (ed. Sayre, D.) 303–317 (Clarendon, Oxford, 1982).
Gunsalus, R. P. & Yanofsky, C. Proc. natn. Acad. Sci. U.S.A. 77, 7117–7121 (1980).
Herzberg, O. & James, M. N. G. Nature 313, 653–659 (1985).
Sundaralingam, M. et al. Science 227, 945–948 (1985).
Babu, Y. S. et al. Nature 315, 37–40 (1985).
Ohlendorf, D. H., Anderson, W. F., Lewis, M., Pabo, C. O. & Matthews, B. W. J. molec. Biol. 169, 757–769 (1983).
Steitz, T. A., Ohlendorf, D. H., McKay, D. B., Anderson, W. F. & Matthews, B. W. Proc. natn. Acad. Sci. U.S.A. 79, 3097–3100 (1982).
Ohlendorf, D. H., Anderson, W. F. & Matthews, B. W. J. molec. Evol. 19, 109–114 (1983).
Pabo, C. O., Krovatin, W., Jeffrey, A. & Sauer, R. T. Nature 298, 441–443 (1982).
Wulff, D. L., Mahoney, M., Shatzman, A. & Rosenberg, M. Proc. natn. Acad. Sci. U.S.A. 81, 555–559 (1984).
Bennett, G. N. & Yanofsky, C. J. molec. Biol. 121, 179–192 (1978).
Arnott, S. & Hukins, D. W. L. Biochem. Biophys. Res. Commun. 47, 1504–1509 (1972).
Calladine, C. R. J. molec. Biol. 161, 343–352 (1982).
Dickerson, R. E. J. molec. Biol. 166, 419–441 (1983).
Matthew, J. B. & Ohlendorf, D. H. J. biol. Chem. 260, 5860–5862 (1985).
Weber, I. T. & Steitz, T. A. Proc. natn. Acad. Sci. U.S.A. 81, 3973–3977 (1984).
Lewis, M. et al. Cold Spring Harb. Symp. quant. Biol. 47, 435–440 (1983).
Ohlendorf, D. H., Anderson, W. F., Fisher, R. G., Takeda, Y. & Matthews, B. W. Nature 298, 718–723 (1982).
Hol, W. G. J., van Duijnen, P. T. & Berendsen, H. J. C. Nature 273, 443–446 (1978).
Oppenheim, D. S., Bennett, G. N. & Yanofsky, C. J. molec. Biol. 144, 133–142 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schevitz, R., Otwinowski, Z., Joachimiak, A. et al. The three-dimensional structure of trp repressor. Nature 317, 782–786 (1985). https://doi.org/10.1038/317782a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/317782a0
- Springer Nature Limited
This article is cited by
-
Directed evolution of a synthetic phylogeny of programmable Trp repressors
Nature Chemical Biology (2018)
-
The structure of trp RNA-binding attenuation protein
Nature (1995)
-
The possible roles of residues 79 and 80 of the Trp repressor from Escherichia coli K-12 in trp operator recognition
Molecular and General Genetics MGG (1995)
-
Study ofl-tryptophan corepressor binding to mutatedE. coli tryptophan repressor proteins by optically detected triplet-state magnetic resonance
Journal of Fluorescence (1994)
-
A mathematical model for the regulation of tryptophan promoter
Korean Journal of Chemical Engineering (1994)