Abstract
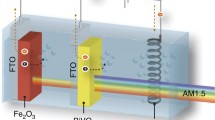
Photoelectrolysis of water by means of semiconductor photo-electrochemical cells has attracted much attention from the point of view of solar energy conversion1–5. Metal oxides such as TiO2, WO3, or Fe2O3 act as stable photoanodes, but they need an external bias for the photodecomposition of water to compensate for their too low surface band energies. We report here that water can be photodecomposed effectively without external bias by using a semiconductor/redox electrolyte/semiconductor junction (SES) as the photoanode.
Similar content being viewed by others
References
Maruska, H. P. & Ghosh, A. K. Solar Energy 20, 443–458 (1978).
Scaife, D. E. Solar Energy 25, 41–54 (1980).
Gerischer, H. Topics Appl. Phys. 31, 115–172 (1979).
Bard, A. J. J. Photochem. 10, 59–75 (1979).
Wrighton, M. S. Accts chem. Res. 12, 303–310 (1979).
Minoura, H., Tsuiki, M. & Oki, T. Ber. Bunsenges. Phys. Chem. 81, 588–592 (1977).
Tomkiewicz, M. J. electrochem. Soc. 126, 1505–1510 (1979).
Wagner, S. & Shay, J. L. Appl. Phys. Lett. 31, 446–447 (1977).
Morisaki, H., Watanabe, T., Iwase, M. & Yazawa, K. Appl. Phys. Lett. 29, 338–340 (1976).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nakato, Y., Takamori, N. & Tsubomura, H. A composite semiconductor photoanode for water electrolysis. Nature 295, 312–313 (1982). https://doi.org/10.1038/295312a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/295312a0
- Springer Nature Limited
This article is cited by
-
Improved water electrolysis using magnetic heating of FeC–Ni core–shell nanoparticles
Nature Energy (2018)
-
Hydrogen generation at irradiated oxide semiconductor–solution interfaces
Journal of Applied Electrochemistry (2007)