Abstract
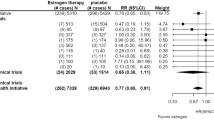
Non-steroidal anti-oestrogens such as tamoxifen, CI 628, nafoxidine and clomiphene, are structurally related synthetic compounds that antagonize the effects of oestrogen on its target tissues1,2, and this activity has led to the use of tamoxifen to treat advanced breast cancer3. All these compounds inhibit the binding of tritiated oestradiol to cytosol from oestrogen target tissues2,4–7, suggesting that anti-oestrogens bind to the oestrogen receptor. This is supported by reports that in the rat uterus7–10 and dimethyl-benz (α)-anthracene (DMBA)-induced rat mammary carcinoma9, oestradiol and anti-oestrogens bind directly to the same number of saturable binding sites. Furthermore, oestrogens and anti-oestrogens are mutually competitive for binding to these sites8,10. It has thus been generally accepted that the anti-oestrogens exert most of their effects through the specific oestrogen receptor. We now report a further high-affinity, anti-oestrogen binding site which may have a role in regulating the effects of non-steroidal anti-oestrogens.
Similar content being viewed by others
References
Katzenellenbogen, B. S. et al. Rec. Prog. Horm. Res. 35, 259–300 (1979).
Jordan, V. C., Dix, D. J., Naylor, K. E., Prestwich, G. & Rowsby, L. J. Tox. envir. Hlth 4, 363–390 (1978).
Mouridsen, H., Palshof, T., Patterson, J. & Battersby, L. Cancer Treat. Rev. 5, 131–141 (1978).
Horwitz, K. B. & McGuire, W. L. J. biol. Chem. 253, 8185–8191 (1978).
Lippman, M., Bolan, G. & Huff, K. Cancer Res. 36, 4595–4601 (1976).
Skidmore, J., Walpole, A. L. & Woodburn, J. J. Endocr. 52, 289–298 (1972).
Sutherland, R. L. & Foo, M. S. Biochem. biophys. Res. Commun. 91, 183–191 (1979).
Katzenellenbogen, B. S., Katzenellenbogen, J. A., Ferguson, E. R. & Krauthammer, N. J. biol. Chem. 253, 697–707 (1978).
Nicholson, R. I., Syne, J. S., Daniel, C. P. & Griffiths, K. Eur. J. Cancer 15, 317–329 (1979).
Capony, F. & Rochefort, H. Mol. cell. Endocr. 11, 181–198 (1978).
Sutherland, R. L. & Simpson-Morgan, M. W. J. Endocr. 65, 319–332 (1975).
Chamness, G. C. & McGuire, W. L. Steroids 26, 538–542 (1976).
Scatchard, G., Ann, N. Y. Acad. Sci. 51, 660–672 (1949).
Murphy, L. C. & Sutherland, R. L. J. Endocr. (in the press).
Sutherland, R. L. & Murphy, L. C. Eur. J. Cancer. (in the press).
Sutherland, R. L. & Foo, M. S. in Non-Steroidal Antioestrogens (eds Sutherland, R. L. & Jordan, V. C.) (Academic, Sydney, in the press).
Jordan, V. C., Collins, M. M., Rowsby, L. & Prestwich, G. J. Endocr. 75, 305–316 (1977).
Binart, N. et al. Biochem. biophys. Res. Commun. 91, 812–818 (1979).
Wakeling, A. E. & Slater, S. R. Cancer Treat. Rep. 63, 1150 (1979).
Faye, J. C., Lasserre, B. & Bayard, F. Biochem. biophys. Res. Commun. 93, 1225–1231 (1980).
Murphy, L. C. et al. in Non-Steroidal Antioestrogens (eds Sutherland, R. L. & Jordan, V. C.) (Academic, Sydney, in the press).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sutherland, R., Murphy, L., Foo, M. et al. High-affinity anti-oestrogen binding site distinct from the oestrogen receptor. Nature 288, 273–275 (1980). https://doi.org/10.1038/288273a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/288273a0
- Springer Nature Limited
This article is cited by
-
Microsomal epoxide hydrolase expression in the endometrial uterine corpus is regulated by progesterone during the menstrual cycle
Journal of Molecular Histology (2010)
-
Ligands of the antiestrogen-binding site induce active cell death and autophagy in human breast cancer cells through the modulation of cholesterol metabolism
Cell Death & Differentiation (2009)
-
Farnesyl-transferase inhibitor R115,777 enhances tamoxifen inhibition of MCF-7 cell growth through estrogen receptor dependent and independent pathways
Breast Cancer Research (2005)