Abstract
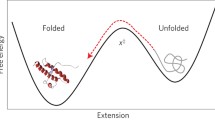
There are two distinct experimental and theoretical problems of protein folding: the thermodynamic issue of characterizing the folded state, and the kinetic question of the path between the unfolded and native states1. Here we consider the second question and present a diffusion–collision–adhesion model for the folding of the α-helical protein myoglobin. In particular, we consider the fast refolding species of the unfolded state and ignore the slow transition between unfolded states that has been attributed to proline isomerization2.
Similar content being viewed by others
References
Creighton, T. E. Prog. Biophys. molec. Biol. 33, 231–297 (1978).
Brandts, J. F., Halvorson, H. R. & Brennan, M. Biochemistry 14, 4953–4963 (1975).
Baldwin, R. L. Protein Folding (ed. Jaenicke, R.) (Elsevier, Amsterdam, 1980).
Karplus, M. & Weaver, D. L. Nature 260, 404–406 (1976).
Karplus, M. & Weaver, D. L. Biopolymers 18, 1421–1437 (1979).
Ptitsyn, O. B. & Rashin, A. A. Biophys. Chem. 3, 1–20 (1975).
Richmond, T. J. & Richards, F. M. J. molec. Biol. 119, 537–555 (1978).
Cohen, F. E., Richmond, T. J. & Richards, F. M. J. molec. Biol. 132, 275–288 (1979).
Shen, L. L. & Hermans, J. Biochemistry 11, 1836–1841 (1972).
Shen, L. L. & Hermans, J. Biochemistry 11, 1845–1849 (1972).
Phillips, D. C. Proc. natn. Acad. Sci. U.S.A. 57, 484–495 (1967).
Hamlin, J. & Zabin, I. Proc. natn. Acad. Sci. U.S.A. 69, 412–416 (1972).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cohen, F., Sternberg, M., Phillips, D. et al. A diffusion–collision–adhesion model for the kinetics of myoglobin refolding. Nature 286, 632–634 (1980). https://doi.org/10.1038/286632a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/286632a0
- Springer Nature Limited