Abstract
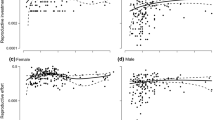
REPRODUCTION is often a lethal or semi-lethal activity, and for iteroparous plants it is often possible to show that reproduction has costs that are expressed in a reduced growth rate and/or an increased death rate1. Attempts have been made to compare life history patterns in flowering plants by measuring the fraction of a plant's annual dry matter production (or calorific value) that is allocated to reproduction (for example refs 2–4). The assumption is that the reproductive parts represent a cost in energy or materials. Clearly mineral nutrients and water must be gained by the reproductive structures from the remainder of the plant, but this is not necessarily true for the energetic and carbon economy of the reproductive structures. Many flowers and fruits are green and a fraction of the energy and carbon might be obtained by direct photosynthesis within these structures. This might be especially important during embryo growth if carpels and other organs that remain attached after flowering are green. In such cases the conventional estimation of reproductive effort (dividing seed weight by plant weight) would be incorrect and comparison of life history patterns and their evolutionary meaning would be invalid. There are reports of significant contributions of in situ photosynthesis in flower and seeds to their growth. Biscoe et al. have estimated that 47% of the carbon required for seed production in barley is provided by photosynthesis of reproductive and immediately adjacent plant structures5. Bazzaz and Carlson have shown that in the annual weed Ambrosia trifida L., net photosynthesis by reproductive structures amounts to 41 and 51%, respectively, of the carbon required for male and female inflorescences6. Here we report an analysis of the carbon budget of reproduction for 15 temperate deciduous trees. The budget was determined by measuring the weight, photosynthesis and dark respiration of flowers or inflorescences from bud break until seed maturity.
Similar content being viewed by others
References
Harper, J. L. Population Biology in Plants (Academic, London, 1977).
Ogden, J. thesis, Univ. Wales (1968).
Abrahamson, W. G. & Gadgil, M. D. Am. Nat. 107, 651–666 (1973).
Gadgil, M. D. & Solbrig, O. T. Am. Nat. 106, 14–31 (1972).
Biscoe, P. V., Gallagher, J. N., Littleton, E. J., Monteith, J. L. & Scott, R. K. J. appl. Ecol. 12, 295–318 (1975).
Bazzaz, F. A. & Carlson, R. W. New Phytol. 82, 223–232 (1979).
Bazzaz, F. A. & Boyer, J. S. Ecology 53, 343–349 (1972).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BAZZAZ, F., CARLSON, R. & HARPER, J. Contribution to reproductive effort by photosynthesis of flowers and fruits. Nature 279, 554–555 (1979). https://doi.org/10.1038/279554a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/279554a0
- Springer Nature Limited
This article is cited by
-
Reproductive compensatory photosynthesis in a semi-arid rangeland bunchgrass
Oecologia (2023)
-
Allocation to male vs female floral function varies by currency and responds differentially to density and moisture stress
Heredity (2017)
-
Leaf vs. inflorescence: differences in photosynthetic activity of grapevine
Photosynthetica (2017)
-
Photosynthetic response of tetraploid and hexaploid wheat to water stress
Photosynthetica (2017)
-
Photosynthetic capacity of the capsule wall and its contribution to carbon fixation and seed yield in castor (Ricinus communis L.)
Acta Physiologiae Plantarum (2016)