Abstract
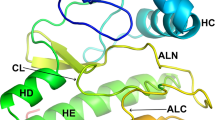
The structure determination of pyruvate kinase shows that each subunit of the tetrameric molecule consists of three domains. The largest of these domains has a remarkable similarity to the structure of triosephosphate isomerase. Another domain shows similarities to many other nucleotide binding proteins. We discuss these similarities and their implications for current arguments on protein taxonomy and evolution.
Similar content being viewed by others
References
Arndt, U. W., Champness, J. N., Phizackerley, R. P. & Wonacott, A. J. J. appl. Crystallogr. 6, 457–463 (1973).
Dayhoff, M. O., Hunt, L. T., McLaughlin, P. J. & Jones, D. D. Atlas of Protein Sequence and Structure 5, 17–30 (1972).
Hartley, B. S. Phil. Trans. R. Soc. B 257, 77–78 (1970).
Richardson, J. S. Proc. natn. Acad. Sci., U.S.A. 73, 2619–2623 (1976).
Sternberg, M. J. E. & Thornton, J. M. J. molec. Biol. 105, 367–382 (1976).
Rossmann, M. G., Moras, D. & Olsen, K. W. Nature 250, 194–199 (1974).
Ohlsson, I., Nordström, B. & Brändén, C-I. J. molec. Biol. 89, 339–354 (1974).
Eventoff, W. & Rossmann, M. G. in CRC Critical Reviews of Biochemistry (ed. Fasman, G. D.), vol. 3, 111–140 (CRC Press, Cleveland, 1975).
Banner, D. W. et al. Nature 255, 609–614 (1975).
Banner, D. W., Bloomer, A. C., Petsko, G. A., Phillips, D. C. & Wilson, I. A. Biochem. biophys. Res. Commun. 72, 146–155 (1976).
Blake, C. C. F. & Evans, P. R. J. molec. Biol. 84, 585–601 (1974).
Bryant, T. N., Watson, H. C. & Wendell, P. L. Nature 247, 14–17 (1974).
Schulz, G. E., Elzinga, M., Marx, F. & Schirmer, R. H. Nature 250, 120–123 (1974).
Steitz, T. A., Fletterick, R. J., Anderson, W. F. & Anderson, C. M. J. molec. Biol. 104, 197–222 (1976).
Stammers, D. K. & Muirhead, H. J. molec. Biol. 112, 309–316 (1977).
Rossmann, M. G. & Argos, P. J. molec. Biol. 105, 75–95 (1976).
Levitt, M. & Chothia, C. Nature 261, 552–558 (1976).
Stammers, D. K. & Muirhead, H. J. molec. Biol. 95, 213–225 (1975).
Rose, I. A. & Rieder, S. V. J. biol. Chem. 234, 1007–1010 (1959).
Rose, I. A. Brookhaven Synp. Biol. 15, 293–309 (1962).
Rose, I. A. J. biol. Chem. 235, 1170–1177 (1960).
Schulz, G. E. & Schirmer, R. H. Nature 250, 142–144 (1974).
Rossmann, M. G. & Argos, P. J. biol. Chem. 250, 7525–7532 (1975).
Rao, S. T. & Rossmann, M. G. J. molec. Biol. 76, 241–256 (1973).
Robinson, J. L. & Rose, I. A. J. biol. Chem. 247, 1096–1105 (1972).
Phillips, D. C., Rivers, P. S., Sternberg, M. J. E., Thornton, J. M. & Wilson, I. A. Biochem. Soc. Trans. 5, 642–647 (1977).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Levine, M., Muirhead, H., Stammers, D. et al. Structure of pyruvate kinase and similarities with other enzymes: possible implications for protein taxonomy and evolution. Nature 271, 626–630 (1978). https://doi.org/10.1038/271626a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/271626a0
- Springer Nature Limited
This article is cited by
-
Comparison of goose-type, chicken-type, and phage-type lysozymes illustrates the changes that occur in both amino acid sequence and three-dimensional structure during evolution
Journal of Molecular Evolution (1985)
-
Methionyl-tRNA synthetase shows the nucleotide binding fold observed in dehydrogenases
Nature (1981)
-
Genes pieced together — exons delineate homologous structures of diverged lysozymes
Nature (1981)
-
Common precursor of lysozymes of hen egg-white and bacteriophage T4
Nature (1981)
-
Characterization ofκ-casein and keratin domains in fibrinogen
Journal of Molecular Evolution (1981)