Abstract
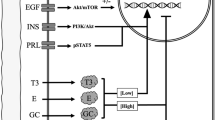
THE activity of arginase in mammary gland increases markedly during lactation1–3. Since lactating mammary gland, unlike liver, lacks other enzymes of the urea cycle3, the function of arginase in this gland is somewhat restricted. Results of earlier studies3,4 have suggested that the enzyme is involved in proline formation, in concert with ornithine aminotransferase, for increased synthesis of milk protein.
Similar content being viewed by others
References
Folley, S. J., and Greenbaum, A. L., Biochem. J., 41, 261–268 (1947).
Greengard, O., Sahib, M. K., and Knox, W. E., Archs Biochem. Biophys., 137, 477–482 (1970).
Yip, M. C. M., and Knox, W. E., Biochem. J., 127, 893–899 (1972).
Mepham, T. B., and Linzell, J. L., Nature, 214, 507–508 (1967).
Russell, D. H., and McVicker, T. A., Biochem. J., 130, 71–76 (1972).
Tabor, H., and Tabor, C. W., Adv. Enzymol., 36, 203–268 (1972).
Oka, T., Science, 184, 78–80 (1974).
Topper, Y. J., Rec. Prog. Hormone Res., 26, 287–308 (1970).
Oka, T., and Perry, J. W., J. biol. Chem., 249, 3586–3591 (1974).
Stockdale, F. E., and Topper, Y. J., Proc. natn. Acad. Sci. U.S.A., 56, 1283–1289 (1966).
Turkington, R. W., Lockwood, D. H., and Topper, Y. J., Biochim. biophys. Acta, 148, 475–480 (1967).
Mills, E. S., and Topper, Y. J., J. Cell Biol., 44, 310–328 (1970).
Oka, T., and Topper, Y. J., J. biol. Chem., 246, 7701–7707 (1971).
Oka, T., and Perry, J. W., J. biol. Chem. (in the press).
Aisbitt, R. P. G., and Barry, J. M., Biochim. biophys. Acta, 320, 610–616 (1973).
Juergens, W. G., Stockdale, F. E., Topper, Y. J., and Elias, J. J., Proc. natn. Acad. Sci. U.S.A., 54, 629–634 (1965).
Schimke, R. T., J. biol. Chem., 239, 3809–3817 (1964).
Vonderhaar, B. K., Owens, I. S., and Topper, Y. J., J. biol. Chem., 248, 467–471 (1973).
Turkington, R. W., and Topper, Y. J., Endocrinology, 79, 175–181 (1966).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
OKA, T., PERRY, J. Arginase affects lactogenesis through its influence on the biosynthesis of spermidine. Nature 250, 660–661 (1974). https://doi.org/10.1038/250660a0
Received:
Issue Date:
DOI: https://doi.org/10.1038/250660a0
- Springer Nature Limited
This article is cited by
-
Linking our understanding of mammary gland metabolism to amino acid nutrition
Amino Acids (2014)
-
Proline and hydroxyproline metabolism: implications for animal and human nutrition
Amino Acids (2011)
-
Regulation of glucose utilization and lipogenesis in adipose tissue of diabetic and fat fed animals: Effects of insulin and manganese
Journal of Biosciences (2003)
-
Role of arginase in the epidermis
Nature (1974)