Abstract
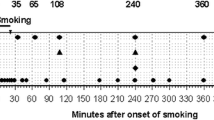
TWO isomers of tetrahydrocannabinol (THC) are found in natural marijuana preparations. 99 per cent is in the form of Δ1(9)-THC, which is very active when taken orally or smoked1,2. A Δ3-THC homologue, synhexyl (pyrahexyl), has been found to be about one-third as active as Δ1-THC, given orally to the same individuals2. The study I report here compared measured doses of Δ1-THC, the Δ3-homologue (synhexyl), and Δ3-THC smoked by the same individuals, using a placebo and blind controls.
Similar content being viewed by others
References
Isbell, H., Gorodetsky, G. W., Jasinski, D., Claussen, U., v. Spulak, F., and Korter, F., Psychopharmacologia, 11, 184 (1967).
Hollister, L. E., Richards, R. K., and Gillespie, H. K., Clin. Pharmacol. Ther., 9, 783 (1968).
Song, Chung Hun, Kanter, S. L., and Hollister, L. E., Res. Comm. Chem. Path. Pharmacol., 1, 375 (1970).
Hollister, L. E., and Gillespie, H. K., Arch. Gen. Psychiat. (in the press).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
HOLLISTER, L. Tetrahydrocannabinol Isomers and Homologues: Contrasted Effects of Smoking. Nature 227, 968–969 (1970). https://doi.org/10.1038/227968a0
Received:
Issue Date:
DOI: https://doi.org/10.1038/227968a0
- Springer Nature Limited
This article is cited by
-
Interactions of cannabinoids with bovine serum albumin
Bioscience Reports (1984)
-
Effects of acute marijuana smoking on pulse rate and mood states in women
Psychopharmacology (1984)