Abstract
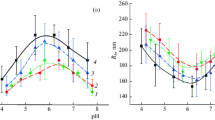
ALTHOUGH several investigations of the infra-red spectrum of collagen in the 2µ region have been reported1, no comment appears to have been made on the fact that the absorption band at 5,150 cm.−1, generally attributed to sorbed water, is dichroic. The extent of this dichroism is shown in Fig. 1 (above), obtained from kangaroo tail tendon in equilibrium with the laboratory atmosphere. In a further spectrum obtained after drying at 100° C. for 3 hr., shown in Fig. 1 (below), the absorption band at 5,150 cm.−1 is virtually eliminated, thus confirming its assignment to sorbed water. Subsequent exposure to the laboratory atmosphere restored the dichroic absorption band at 5,150 cm.−1.
Similar content being viewed by others
References
Ambrose, E. J., and Elliott, A., Proc. Roy. Soc., A, 206, 206 (1951). Sutherland, G. B. B. M., Tanner, K. N., and Wood, D. L., J. Chem. Phys., 22, 1621 (1954). Hecht, K. T., and Wood, D. L., Proc. Roy. Soc., A, 235, 174 (1956).
Fraser, R. D. B., J. Chem. Phys. (in the press).
Crick, F. H. C., and Kendrew, J. C., “Adv. in Protein Chem.”, 12, 133 (1957).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
FRASER, R., MACRAE, T. Possible Role of Water in Collagen Structure. Nature 183, 179–180 (1959). https://doi.org/10.1038/183179a0
Issue Date:
DOI: https://doi.org/10.1038/183179a0
- Springer Nature Limited
This article is cited by
-
Stabilization of the collagen structure by hydroxyproline residues
Proceedings of the Indian Academy of Sciences - Section A (1975)
-
Grenzflächeneffekte und Proteinkonformation von Faserkeratinen
Fortschrittsberichte über Kolloide und Polymere (1971)
-
Infra-red Examination of the Association of Cornea Proteins with Water
Nature (1960)