Abstract
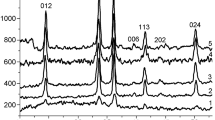
THE chromium–aluminium oxide catalyst system is one most commonly used in the aromatization of paraffinic hydrocarbons. In practice, mixtures containing up to 20 per cent chromium oxide are used, this being regarded as the region of greatest catalytic activity. The work of Eischens and Selwood1 on this catalyst system established the close relation between the activity, magnetic susceptibility, and state of dispersion of the chromium oxide on the alumina. It was shown that at low chromium oxide content, the chromium oxide aggregates into clusters or islands which build up into two and even three layers in thickness, and which have high magnetic susceptibility and high catalytic activity.
Similar content being viewed by others
References
Eischens, R. P., and Selwood, P. W., J. Amer. Chem. Soc., 70, 2271 (1948).
Hawkins, P. J., and Janz, G. J., J. Chem. Soc., 1485 (1949).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
JANZ, G., HAWKINS, P. Activity of the Chromium-Aluminium Oxide Catalyst System in the Nitrile-Diene and n-Heptane Cyclizations. Nature 168, 387–388 (1951). https://doi.org/10.1038/168387a0
Published:
Issue Date:
DOI: https://doi.org/10.1038/168387a0
- Springer Nature Limited