Abstract
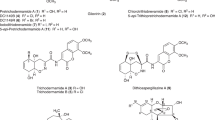
GLIOTOXIN, a potent fungicide isolated from culture filtrates of Gliocladium fimbriatum, Trichoderma viride and other organisms1, has been extensively examined by J. R. Johnson and his colleagues2, who ascribe to it the provisional structure (I). The presence of the pyrazinoindole nucleus and of the disulphide grouping appear to be established with reasonable certainty. The points of attachment of the disulphide grouping to the pyrazinoindole nucleus, on the other hand, merit further confirmation. Treatment of gliotoxin with methanolic potassium hydroxide followed by acidification of the reaction mixture gives a sulphur-containing product, m.p. 188°, which appears to have the molecular formula C11H8ON2S and for which the thiohydantoin structure (II) has been suggested3.
Similar content being viewed by others
References
Weindling and Emerson, Phytopath., 26, 1068 (1936). Weindling, ibid., 24, 1153 (1934); 31, 991 (1941). Brian, Nature, 154, 667 (1944). Brian and Hemming, Ann. App. Biol., 32, 214 (1945). Waksman and Geiger, J. Bact., 47, 391 (1944). Menzel, Winter-Steiner and Hoogerheide, J. Biol. Chem., 152, 419 (1944). Glister and Williams, Nature, 153, 651 (1944). Brian, Trans. Brit. Mycol. Soc., 29, 211 (1946). Stanley, Aust. J. Sci., 6, 151 (1944). Stanley and Mills, Aust. J. Exp. Biol. and Med. Sci., 24, 133 (1946).
J. Amer. Chem. Soc., 65, 2005 (1943); 66, 614, 617, 619 (1944); 67, 423, 1736 (1945); 69, 2364 (1947).
Dutcher, Johnson and Bruce, J. Amer. Chem. Soc., 67, 1736 (1945).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ELVIDGE, J., SPRING, F. Synthesis of a Sulphur-containing Degradation Product from Gliotoxin. Nature 163, 94 (1949). https://doi.org/10.1038/163094a0
Published:
Issue Date:
DOI: https://doi.org/10.1038/163094a0
- Springer Nature Limited