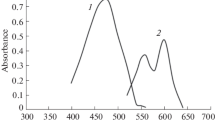
Abstract
THE platinum potential of solutions containing biological oxidation-reduction agents can be measured when exposed to air by means of a relatively simple technique1.
Similar content being viewed by others
References
Bezssonoff, N., and Woloszyn, M., Bull. Soc. Chim. Biol., 21, 208 (1939).
Bezssonoff and Woloszyn, loc. cit. The pH stability of the ascorbic acid solutions was ascertained by means of an antimony electrode system.
Heintz, C.R. Acad. Sci., 208, 1893 (1939).
Borsook, H., and Keighley, G., Proc. Nat. Acad. Sci., 19, 1875 (1933). Wurmser, R., Actual. Sci. et Indus., 244, 56 (1935). Ball, E., J. Biol. Chem., 118, 219 (1937).
According to the potentiometric measurements of Vles, Reiss and Deloyers, C.R. Soc. Biol, 108, 37 (1931). Outevskaya, Bull. Biol. et Med. Exper. U.R.S.S., 5, 226 (1938). Rafalkes, Tzoutzoul-kovskaya et Chneerson, Bull. Soc. et Med. Exper. U.R.S.S., 5, 191 (1938).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BEZSSONOFF, N., WOLOSZYN, M. Platinum Potential of Vitamin C Solutions in Contact with Molecular Oxygen. Nature 144, 289–290 (1939). https://doi.org/10.1038/144289a0
Issue Date:
DOI: https://doi.org/10.1038/144289a0
- Springer Nature Limited