Abstract
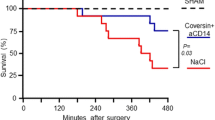
Sepsis in humans is a difficult condition to treat and is often associated with a high mortality rate. In this study, we induced sepsis in rats using cecal ligation and puncture (CLP). In rats depleted of the complement factor C3, CLP led to very short survival times (about 4 days). Of the rats that underwent CLP ('CLP rats') that were C3-intact and treated with preimmune IgG, most (92%) were dead by 7 days. Blood neutrophils from these rats contained on their surfaces the powerful complement activation product C5a. This group had high levels of bacteremia, and their blood neutrophils when stimulated in vitro had greatly reduced production of H2O2, which is known to be essential for the bactericidal function of neutrophils. In contrast, when companion CLP rats were treated with IgG antibody against C5a, survival rates were significantly improved, levels of bacteremia were considerably reduced, and the H2O2 response of blood neutrophils was preserved. Bacterial colony-forming units in spleen and liver were very high in CLP rats treated with preimmune IgG and very low in CLP rats treated with IgG antibody against C5a, similar to values obtained in rats that underwent 'sham' operations (without CLP). These data indicate that sepsis causes an excessive production of C5a, which compromises the bactericidal function of neutrophils. Thus, C5a may be a useful target for the treatment of sepsis.




Similar content being viewed by others
References
Bone, R.C. Toward a theory regarding the pathogenesis of the systemic inflammatory response syndrome: what we do and do not know about cytokine regulation. Crit. Care Med. 24, 163–172 (1996).
Nichols, R. et al. Risk of infection after penetrating abdominal trauma. N. Engl. J. Med. 311, 1065–1070 (1984).
Border, J.R. et al. The gut origin sepsis state in blunt multiple trauma (ISS=40) in the ICU. Ann. Surg. 206, 427–448 (1987).
Dinarello, C.A., Gelfand, J.A. & Wolff, S.M. Anticytokine strategies in the treatment of the systemic inflammatory response syndrome. J. Am. Med. Assoc. 269, 1829–1835 (1993).
Deitch, E.A. Animal models of sepsis and shock: a review and lessons learned. Shock 9, 1–11 (1997).
Wichtermann, K.A., Bauer, A.E. & Chaudry, I.H. Sepsis and shock – a review of laboratory models and a proposal. J. Surg. Res. 29, 189–201 (1980).
Nakae, H., Endo, S., Inada, K. & Yoshida, M. Chronological changes in the complement system in sepsis. Jpn. J. Surg. 26, 225–229 (1996).
Quezado, Z.M. et al. The third component of complement protects against Escherichia coli endotoxin-induced shock and multiple organ failure. J. Exp. Med. 179, 569–578 (1994).
Kubens, B.S. & Opferkuch, W. in The Complement System (eds. Rother, K. & Till, G.O.) 469–487 (Springer-Verlag, Berlin, 1988).
Koehl, J. & Bitter-Suermann, D. in Anaphylatoxins. Complement in Health and Disease (eds. Whaley, K., Loos, M. & Weiler, J.M.) 299–324 (Kluwer Academic, London, 1993).
Klebanoff, S.J. Antimicrobial mechanisms in neutrophilic polymorphonuclear leukocytes. Semin. Hematol. 12, 117–142 (1975).
Elsbach, P. & Weiss, J. Oxygen-dependent and oxygen-independent mechanisms of microbicidal activity of neutrophils. Immunol. Lett. 11, 159–163 (1983).
Wessels, M.R. Studies of group B streptococcal infection in mice deficient in complement component C3 or C4 demonstrate an essential role for complement in both innate and acquired immunity. Proc. Natl. Acad. Sci. USA 92, 11490–11494 (1995).
Fischer, M.B. et al. Increased susceptibility to endotoxin shock in complement C3- and C4-deficient mice is corrected by C1 inhibitor replacement. J. Immunol. 159, 976–982 (1997).
May, J.E., Kane, M.A. & Frank, M.M. Host defense against bacterial endotoxemia–contribution of the early and late components of complement to detoxification. J. Immunol. 109, 893–895 (1972).
Schumacher, W.A., Fantone, J.C., Kunkel, S.E., Webb, R.C. & Lucchesi, B.R. The anaphylatoxins C3a and C5a are vasodilators in the canine coronary vasculature in vitro and in vivo. Agents Actions 34, 345–349 (1991).
Ward, P.A. & Becker, E.L. The deactivation of rabbit neutrophils by chemotactic factor and the nature of activatable esterase. J. Exp. Med. 127, 693–709 (1968).
Solomkin, J.S., Jenkins, M.K., Nelson, R.D., Chenoweth, D. & Simmons, R.L. Neutrophil dysfunction in sepsis. II. Evidence for the role of complement activation products in cellular deactivation. Surgery 90, 319–327 (1981).
Mulligan, M.S. et al. Requirement and role of C5a in acute inflammatory lung injury in rats. J. Clin. Invest. 98, 503–512 (1996).
Hill, J.H. & Ward, P.A. The phlogistic role of C3 leukotactic fragments in myocardial infarcts of rats. J. Exp. Med. 133, 885–900 (1971).
Alexander, J.W. et al. A comparison of immunologic profiles and their influence on bacteremia in surgical patients with a high risk of infection. Surgery 86, 84–104 (1979).
Mohr M et al. Effects of anti-C5a monoclonal antibodies on oxygen use in a porcine model of severe sepsis. Eur. J. Clin. Invest. 28, 227–234 (1998).
Stevens, J.H. et al. Effects of anti-C5a antibodies on the adult respiratory distress syndrome in septic primates. J. Clin. Invest. 77, 1812–1816 (1986).
Johnson, K.J. & Ward, P.A. Protective function of C6 in rabbits treated with bacterial endotoxin. J. Immunol. 106, 1125–1127 (1971).
Höpken, U.E., Lu, B., Gerard, N.P. & Gerard, C. The C5a chemoattractant receptor mediates mucosal defense to infection. Nature 383, 86–89 (1996).
Naik, N., Giannini, E., Brouchon, L. & Boulay, F. Internalization and recycling of the C5a anaphylatoxin receptor: evidence that the agonist-mediated internalization is modulated by phosphorylation of the C-terminal domain. J. Cell Science. 110, 2381–2390 (1997).
Giannini, E. & Boulay, F. Phosphorylation, dephosphorylation, and recycling of the C5a receptor in differentiated HL60 cells. J. Immunol. 154, 4055–4064 (1995).
Bock, D. et al. The C terminus of the human C5a receptor (CD88) is required for normal ligand-dependent receptor internalization. Eur. J. Immunol. 27, 1522–1529 (1997).
Van Epps, D.E., Simpson, S., Bender, J.G. & Chenoweth, D.E. Regulation of C5a and formyl peptide receptor expression on human polymorphonuclear leukocytes. J Immunol 144, 1062–1068 (1990).
Van Epps, D.E., Simpson, S.J. & Johnson, R. Relationship of C5a receptor modulation to the functional responsiveness of human polymorphonuclear leukocytes to C5a. J. Immunol. 150, 246–252 (1993).
Czermak, B.J. et al. Mechanisms of enhanced lung injury during sepsis. Am J Path 154, 1057–1065 (1999).
Olson, L.M., Moss, G.S., Baukus, O. & Das Gupta, T.K. The role of C5 in septic lung injury. Ann. Surg. 202, 771–776 (1985).
Rothermel, E., Rolf, O., Götze, O. & Zwirner, J. Nucleotide and corrected amino acid sequence of the functional recombinant rat anaphylatoxin C5a. Biochim. Biophys. Acta 1351, 9–12 (1997).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Czermak, B., Sarma, V., Pierson, C. et al. Protective effects of C5a blockade in sepsis. Nat Med 5, 788–792 (1999). https://doi.org/10.1038/10512
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/10512
- Springer Nature America, Inc.
This article is cited by
-
Vitamin D receptor (VDR) on the cell membrane of mouse macrophages participates in the formation of lipopolysaccharide tolerance: mVDR is related to the effect of artesunate to reverse LPS tolerance
Cell Communication and Signaling (2023)
-
During Sepsis and COVID-19, the Pro-Inflammatory and Anti-Inflammatory Responses Are Concomitant
Clinical Reviews in Allergy & Immunology (2023)
-
Cronobacter sakazakii Infection in Early Postnatal Rats Impaired Contextual-Associated Learning: a Putative Role of C5a-Mediated NF-κβ and ASK1 Pathways
Journal of Molecular Neuroscience (2021)
-
Complement as driver of systemic inflammation and organ failure in trauma, burn, and sepsis
Seminars in Immunopathology (2021)
-
Vitamin lipid nanoparticles enable adoptive macrophage transfer for the treatment of multidrug-resistant bacterial sepsis
Nature Nanotechnology (2020)