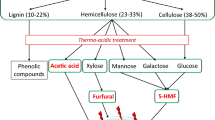
Candida shehatae
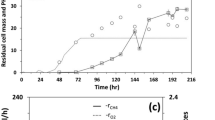
cells pre-grown on D-xylose simultaneously consumed mixtures of D-xylose and D-glucose, under both non-growing (anoxic) and actively growing conditions (aerobic), to produce ethanol. The rate of D-glucose consumption was independent of the D-xylose concentration for cells induced on D-xylose. However, the D-xylose consumption rate was approximately three times lower than the D-glucose consumption rate at a 50% D-glucose: 50% D-xylose mixture. Repression was not observed (substrate utilization rates were approximately equal) when the percentage of D-glucose and D-xylose was changed to 22% and 78%, respectively. In fermentations with actively growing cells (50% glucose and D-xylose), ethanol yields from D-xylose increased, the % D-xylose utilized increased, and the xylitol yield was significantly reduced in the presence of D-glucose, compared to anoxic fermentations (YETOH,xylose = 0.2–0.40 g g−1, 75–100%, and Yxylitol = 0–0.2 g g−1 compared to YETOH,xylose = 0.15 g g−1, 56%, Yxylitol = 0.51 g g−1, respectively). To increase ethanol levels and reduce process time, fed-batch fermentations were performed in a single stage reactor employing two phases: (1) rapid aerobic growth on D-xylose (μ = 0.32 h−1) to high cell densities; (2) D-glucose addition and anaerobic conditions to produce ethanol (YETOH,xylose = 0.23 g g−1). The process generated high cell densities, 2 × 109 cells ml−1, and produced 45–50 g L−1 ethanol within 50 h from a mixture of D-glucose and D-xylose (compared to 30 g L−1 in 80 h in the best batch process). The two-phase process minimized loss of cell viability, increased D-xylose utilization, reduced process time, and increased final ethanol levels compared to the batch process.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received 23 February 1998/ Accepted in revised form 15 July 1998
Rights and permissions
About this article
Cite this article
Kastner, J., Jones, W. & Roberts, R. Ethanol fermentation of mixed-sugars using a two-phase, fed-batch process: method to minimize D-glucose repression of Candida shehatae D-xylose fermentations. J Ind Microbiol Biotech 22, 65–70 (1999). https://doi.org/10.1038/sj.jim.2900608
Issue Date:
DOI: https://doi.org/10.1038/sj.jim.2900608