Abstract
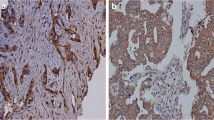
Expression and clinical relevance of p-glycoprotein (p-gp) were evaluated in 31 cases of locally advanced breast cancer and 9 cases involving inflammatory breast cancer after induction chemotherapy. The de novo p-gp expression rate was 26% and increased up to 58% (p = 0.03) with the FAC (5-fluorouracil, adriamycin, cyclophosphamide) regimen. Although more clinically complete responders were found in the secondary p-gp negative group (p = 0.02), this difference was not found in pathological tumor response. Moreover, as the grade of the secondary p-gp expression increased, the chemotherapeutic effect decreased, suggesting an inverse relationship between p-gp expression and drug effect (p = 0.04). When we subgrouped the patients into 4 groups using these two parameters, p-gp negative patients presenting with a high drug effect showed a low recurrence rate (p = 0.05) and marginal survival benefits (p = 0.09) as opposed to patients with a low drug effect. But in p-gp positive groups, the recurrence rate was the same between the two groups regardless of the drug effect. Thus, in the p-gp negative patient with a high drug effect, adjuvant chemotherapy with the same regimen as induction chemotherapy may induce more prognostically favorable results. Therefore, clinical application of the secondary p-gp detection can be used as an intermediate endpoint in evaluating drug response for an induction regimen.
Similar content being viewed by others
References
Zucali R, Uslenghi C, Kenda R, Bonadonna G: Natural history and survival of breast cancer treated with radiotherapy followed by radical mastectomy. Cancer 37: 1422–1431, 1976
Bruckman J, Harris J, Levene M, Chaffey J, Hellman S: Results of treating stage III carcinoma of breast by primary radiation therapy. Cancer 43: 985–993, 1979
Chu A, Wood W, Doucette A: Inflammatory breast carcinoma treated by radical radiotherapy. Cancer 45: 2730–2737, 1980
Balawajder I, Antich PP, Boland J: An analysis of the role of radiotherapy alone in combination with chemotherapy and surgery in the management of advanced breast carcinoma. Cancer 51: 574–580, 1983
Bedwinek J, Rao DV, Perez C, Lee J, Finberg B: Stage III and localized stage IV breast cancer: irradiation alone versus irradiation plus surgery. Int J Radiat Oncol Biol Phys 8: 31–36, 1982
Grohn P, Heinonen E, Klefstrom P, Tarkkanen J: Adjuvant post-operative radiotherapy, chemotherapy and immunotherapy in stage III breast cancer. Cancer 54: 670–674, 1984
DeLena M, Zucali R, Viganotti G, Valagussa P, Bonadonna G: Combined chemotherapy-radiotherapy approach in locally advanced (T3b-T4) breast cancer. Cancer Chemother Pharmacol 1: 53–59, 1978
Hortobagyi GN, Ames GR, Buzdar FC, Kau SW, McNeese MD, Paulus D, Hug V, Holmes FA, Rousdahl MM, Fraschini G, McBrides CM, Martin RG, Montague E: Management of stage III primary breast cancer with primary chemotherapy, surgery and radiation therapy. Cancer 62: 2507–2516, 1988
Bonadonna G, Veronesi U, Brambilla C, Ferrari L, Luini A, Gueco M, Bartoli C, Yoldi GC, Zucali R, Rilke F, Andreola S, Silvestrini R, Di Fronzo G, Valagussa P: Primary chemotherapy to avoid mastectomy with diameters of three centimeters or more. J Natl Cancer Inst 82: 1539–1545, 1990
Bonadonna G, Valagussa P, Brambilla C, Ferrari L: Pre-operative chemotherapy in operable breast cancer. Lancet 341: 1485, 1993
Feldman LD, Hortobagyi GN, Buzdar AV, Ames FC, Blumenschein GR: Pathological assessment of response to induction chemotherapy in breast cancer. Cancer Res 46: 2578–2581, 1986
Schwartz GF, Cantor RI, Biermann WA: Neoadjuvant chemotherapy before definitive treatment for stage III carcinoma of the breast. Arch Surg 122: 1430–1434, 1987
McCready DR, Hortobagyi GN, Kau SW, Smith TL, Buzdar AU, Balch CM: The prognostic significance of lymph node metastasis after preoperative chemotherapy for locally advanced breast cancer. Arch Surg 124: 21–25, 1985
Fojo AT, Ueda K, Slamon DJ, Poplack DG, Gottesman MM, Pastan I: Expression of a multi-drug resistance gene in human tumors and tissues. Proc Natl Acad Sci USA 84: 265–269, 1987
Dalton WS, Grogan TM, Meltzer PS, Scheper RJ, Durie BGM, Taylor CW, Miller TP, Salmon SE: Drug resistance in multiple myeloma and non-Hodgkin's lymphoma: Detection of p-glycoprotein and potential circumvention by addition of verapamil to chemotherapy. J Clin Oncol 7: 415–424, 1989
Chan HSL, Thorner PS, Haddad G, Ling V: Immunohistochemical detection of p-glycoprotein: prognostic correlation in soft tissue sarcoma of childhood. J Clin Oncol 8: 689–704, 1990
Ro JS, Sahin A, Ro JY, Fristsche H, Hortobagyi G, Blick M: Immunohistochemical analysis of p-glycoprotein expression correlated with chemotherapy resistance in locally advanced breast cancer. Hum Pathol 21: 787–791, 1990
Verelle P, Missonnier F, Fonck Y, Feillel V, Dionet C, Kwiatkowski F, Plagne R, Chassagne J: Clinical relevance of immunohistochemical detection of multidrug resistance p-glycoprotein in breast carcinoma. J Natl Cancer Inst 83: 111–116, 1991
Linn SC, Giaccone G, van Diest PJ, Blokhuis WMD, van der Valk P, van Kalken CK, Kuiper CM, Pinedo HM, Baak JPA: Prognostic relevance of p-glycoprotein expression in breast cancer. Ann Oncol 6: 679–685, 1995
Seymour L, Bezwoda WR, Dansey RD: P-glycoprotein immunostaining correlates with ER and with high Ki67 expression but fails to predict anthracycline resistance in patients with advanced breast cancer. Breast Cancer Res Treat 36: 61–69, 1995
Wu YF, Zhen ZA, Shao JK, Zhen HD, Han ZH: Histopathologic effect of preoperative chemotherapy using 5-FU fat emulsion in gastric cancer. J Surg Oncol 29: 50–53, 1985
Sheper RJ, Brakkee JGP, Quak JJ, Shoot EV, Balm AJM, Meijer CJLM, Broxterman MJ, Kuiper CH, Lanklema J, Pinedo HM: Monoclonal antibody JSB-1 detects a highly conserved epitope on the p-glycoprotein associated with multidrug resistance. Int J Cancer 42: 389–394, 1988
Deuchars KL, Ling V: P-glycoprotein and multidrug resistance in cancer chemotherapy. Semin Oncol 16: 156–165, 1989
Merkel DE, Fuqua SAW, Tandon AK, Hill SM, Buzdar AU, MacGuire WL: Electrophoretic analysis of 248 clinical breast cancer specimens for p-glycoprotein overexpression or gene amplification. J Clin Oncol 7: 1129–1136, 1989
Salmon SE, Grogan TM, Miller T, Scheper R, Dalton WS: Prediction of doxorubicin resistance in vitro in myeloma, lymphoma, and breast cancer by p-glycoprotein staining. J Natl Cancer Inst 81: 696–701, 1989
Wishart GC, Plumb JA, Going JJ, McNicol AM, McArdle CS, Tsuruo T, Kage SB: P-glycoprotein expression in primary breast cancer detected by immunocytochemistry with two monoclonal antibodies. Br J Cancer 62: 758–761, 1990
Epstein J, Xiao H, Oba BK: P-glycoprotein expression in plasma-cell myeloma is associated with resistance to VAD. Blood 74: 913–917, 1989
Ma DDF, Davey RA, Harman DH, Isbister JP, Scurr RD, Mackertich SM, Dowden G, Bell DR: Detection of a multidrug resistant phenotype in acute non-lymphoblastic leukemia. Lancet 1: 135–137, 1987
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chung, H.C., Rha, S.Y., Kim, J.H. et al. P-glycoprotein: The intermediate end point of drug response to induction chemotherapy in locally advanced breast cancer. Breast Cancer Res Treat 42, 65–72 (1997). https://doi.org/10.1023/A:1005739525196
Issue Date:
DOI: https://doi.org/10.1023/A:1005739525196