Abstract
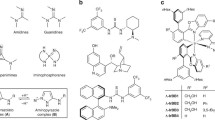
We analyze the merits of an approach to the design of enantioselective catalysts that relies on constraining an anchored organometallic complex at the inner walls of mesoporous silica. Two distinct metal centers {Rh(I) and Pd(II)}, three distinct diamino ligands {(S)-(-)-2-aminomethyl-1-ethylpyrrolidine, (AMP), (S)-(+)-(2-pyrrolidinyl-methyl)pyrrolidine, (PMP), and (1R,2R)-(+)-1,2-diphenylethylenediamine, (DED)}, two distinct methods of anchoring (a covalent and a noncovalent one), and two specific examples of enantioselective hydrogenation (the asymmetric hydrogenation of the C=C bond in E-α-phenylcinnamic acid and the asymmetric hydrogenation of the C=O bond in methyl benzoylformate) have been investigated. Values of enantiomeric excess (ee) ranging from a low of 66 to a high of 93 have been achieved by this approach. Moreover, the catalysts are robust and stand up well to recycling.
Similar content being viewed by others
References
G.J. Hutchings, Chem. Commun. (1999) 301.
R. Noyori, Angew. Chem. Int. Ed. 41 (2002) 2008.
B.K. Sharpless, Angew. Chem. Int. Ed. 41 (2002) 2024.
H.U. Blaser, Chem. Commun. (2003) 293.
M. Shibasaki, M. Kanai and K. Funabashi, Chem. Commun. (2002) 1989.
T. Burgi and A. Baiker, J. Am. Chem. Soc. 120 (1998) 12920.
T.P. Yoon and E.N. Jacobsen, Science 299 (2003) 1691.
D.G. Blackmond, Acc. Chem. Res. 33 (2000) 402.
J.S. Johnson and D.A. Evans, Acc. Chem. Res. 33 (2000) 325.
H. Brunner and W. Zettlmeier, (eds.), Handbook of Enantioselective Catalysis (Wiley-VCH, Weinheim, 1993).
F.H. Arnold, Nature 409 (2001) 253.
M.T. Reetz, M. Rentzsch, A. Pletsch and M. Maywald, Chimica 56 (2002) 721.
G.M. Schwab and L. Rudolph, Naturwissenshaft 20 (1932) 362.
P.B. Wells and A.G. Wilkinson, Top. Catal. 5 (1998) 39.
M. von Arx, T. Mallat and A. Baiker, Top. Catal. 19 (2002) 75.
A.F. Carley, M.K. Rajumon, M.W. Roberts and P.B. Wells, J. Chem. Soc. Faraday Trans. 91 (1995) 2167.
J.M. Thomas, Angew. Chem. Int. Ed. 28 (1989) 1079.
X. Li, R.P.K. Wells, P.B. Wells and G.J. Hutchings, Catal. Lett. (2003), in press.
J.D. Horvath and A.J. Gellman, Top. Catal. (2003), in press.
G.S. Attard and A. Ahmadi, Langmuir 15 (1999) 2420.
S. Feast, D. Bethell, P.C.B. Page, F. King, C.H. Rochester, M.R.H. Siddiqui, D.J. Willock and G.J. Hutchings, J. Chem. Soc. Chem. Commun. (1995) 2409.
S. Taylor, T. Gullick, N. Galea, P. McMorn, D. Bethell, P.C.B. Page, F.E. Hancock, F. King, D.J. Willock and G.J. Hutchings, Top. Catal. (2003), in press.
B.F.G. Johnson, S.A. Raynor, D.S. Shephard, T. Mashmeyer, J.M. Thomas, G. Sankar, S. Bromley, R. Oldroyd, L. Gladden and M.D. Mantle, Chem Commun. (1999) 1167.
S.A. Raynor, J.M. Thomas, R. Raja, B.F.G. Johnson, R.G. Bell and M.D. Mantle, Chem. Commun. (2000) 1925.
R. Raja, J.M. Thomas, M.D. Jones, B.F.G. Johnson and D.E.W. Vaughan, (2003), submitted.
J.M. Thomas Angew. Chem. Int. Ed. 38 (1999) 3588.
J. Rouzard, M.D. Jones, R. Raja, B.F.G. Johnson, J.M. Thomas and M.J. Duer, Helv. Chim. Acta. 86 (2003) 1753.
M.D. Jones, R. Raja, J.M. Thomas, B.F.G. Johnson, D.W. Lewis, J. Rouzard and K.D.M. Harris, Angew. Chem. Int. Ed. (2003) in press.
D.J. Cole-Hamilton, Science 299 (2003) 1702.
J.M. Thomas, Nature 368 (1994) 289.
C.T. Kresge, M.E. Leonowicz, W.J. Roth, J.C. Vartuli and J.S. Beck, Nature 359 (1992) 710.
G.D. Stucky, Nature 368 (1994) 317.
W.Z. Zhou, H.M.A. Hunter, P.A. Wright, Q.F. Ge and J.M. Thomas, J. Phys. Chem. B 102 (1998) 6933.
J.M. Thomas, T. Maschmeyer, B.F.G. Johnson and D.S. Shephard, J. Mol. Catal. A 141 (1999) 139.
J.M. Thomas, R. Raja, B.F.G. Johnson, S. Hermans, M.D. Jones and T. Khimyak, Ind. Eng. Chem. Res. 42 (2003) 1563.
J.M. Thomas, B.F.G. Johnson, R. Raja, G. Sankar and P.A. Midgley, Acc. Chem. Res. 36 (2003) 20.
F.M. de Rege, D.K. Morita, K.C. Ott, W. Tumas, and R.D. Broene, R.D. Chem. Commun. (2000) 1797.
R. Raja, J.M. Thomas, B.F.G. Johnson and M.D. Jones, German Patents. DE 10305946 and DE 10305943 (filed 12 Feb., 2003).
T. Maschmeyer, Oral report to the Davy Faraday Laboratory (March, 1995) (unpublished).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jones, M.D., Raja, R., Meurig Thomas, J. et al. A New Approach to the Design of Heterogeneous Single-Site Enantioselective Catalysts. Topics in Catalysis 25, 71–79 (2003). https://doi.org/10.1023/B:TOCA.0000003099.40587.3a
Issue Date:
DOI: https://doi.org/10.1023/B:TOCA.0000003099.40587.3a