Abstract
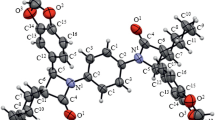
Reformatsky reactions of methyl 1-bromocyclohexanecarboxylate and methyl 1-bromocyclo-pentanecarboxylate with 2-aryl-2-oxoacetaldehydes involve both carbonyl groups of the latter and result in formation of 3a-aryl-3,3 : 6,6-bis(pentamethylene)- and 3a-aryl-3,3 : 6,6-bis(tetramethylene)tetrahydrofuro-[3,2-b]furan-2,5-diones. The reaction with 2-(2,4-dimethylphenyl)-2-oxoacetaldehyde gives acyclic products, methyl 1-[1-hydroxy-2-(2,4-dimethylphenyl)-2-oxoethyl]cyclohexanecarboxylate and methyl 1-[1-hydroxy-2-(2,4-dimethylphenyl)-2-oxoethyl]cyclopentanecarboxylate, while with benzil methyl 1-(4-hydroxy-1-oxo-3,4-diphenyl-2-oxaspiro[4.5]dec-3-yl)cyclohexanecarboxylate and methyl 1-(4-hydroxy-1-oxo-3,4-diphenyl-2-oxaspiro[4.4]non-3-yl)cyclopentanecarboxylate are obtained.
Similar content being viewed by others
REFERENCE
Shchepin, V.V., Fotin, D.V., Nedugov, A.N., Fotin, V.V., and Shurov, S.N., Russ. J. Org. Chem., 2002, vol. 38, p. 256.
Rights and permissions
About this article
Cite this article
Kirillov, N.F., Shchepin, V.V. & Vedernikova, L.A. Reformatsky Reaction of Methyl 1-Bromocycloalkanecarboxylates with α-Dicarbonyl Compounds. Russian Journal of Organic Chemistry 40, 953–956 (2004). https://doi.org/10.1023/B:RUJO.0000045183.34092.05
Issue Date:
DOI: https://doi.org/10.1023/B:RUJO.0000045183.34092.05