Abstract
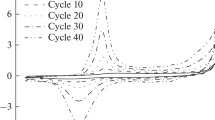
The electrochemical oxidation–reduction of films of polyaniline is studied in the presence of various counteranions. The differential cyclic voltabsorptometric (DCVA) curves (dA/dt relative to potential, where A is the optical absorption) are recorded at characteristic wavelengths corresponding to individual spectrum components previously isolated with the Alentsev–Fok method. The DCVA curves for aqueous solutions of HCl, HClO4, and H2SO4 at different potential scan rates are compared to traditional cyclic voltammograms. For the DCVA curves obtained at 665 nm discovered is one broad peak of dA/dt in the region of potentials between the first and second stages of polyaniline oxidation ( 0.6 V vs. Ag/AgCl). An assumption is made about a chemical nature of the process of generation of absorption in this spectral region, which, more likely than not, is connected with the emergence of dimers of radical cations of polyaniline. It is discovered that the electrochemical processes in the region of potentials that correspond to the first stage of oxidation give rise to variations in the absorption inside several regions of spectrum: 435 nm (radical cations) and 755 nm (localized polarons). An assumption is made that the first peak of the current corresponds to several successive processes that occur in a polyaniline molecule in the course of oxidation as well as to the existence of heterogeneous regions where the generation of localized polarons proceeds at different rates.
Similar content being viewed by others
REFERENCES
Heineman, W.R., Hawkridge, F.M., and Blount, H.N., Electroanalytical Chemistry, Bard, A.J., Ed., New York: Marcel Dekker, 1984, vol. 13, p. 1.
Kuzmany, H. and Sariciftci, N.S., Synth. Met., 1987, vol. 18, p. 353.
Kalaji, M. and Peter, L.M., J. Chem. Soc., Faraday Trans., 1991, vol. 87, p. 853.
Malinauskas, A. and Holze, R., Synth. Met., 1998, vol. 97, p. 31.
Aoki, K. and Teragishi, Y., J. Electroanal. Chem., 1998, vol. 441, p. 25.
Aoki, K., Edo, T., and Cao, J., Electrochim. Acta, 1998, vol. 43, p. 285.
Stilwell, D.E. and Park, S.-M., J. Electrochem. Soc., 1989, vol. 136, p. 427.
Lapkowski, M., Bull. Electrochem., 1989, vol. 5, p. 792.
Gazotti, W.A., Jannini, M.J.D.M., De Torresi, S.I.C., and De Paoli, M.A., J. Electroanal. Chem., 1997, vol. 440, p. 193.
Visy, C., Krivan, E., and Peintler, G., J. Electroanal. Chem., 1999, vol. 462, p. 1.
Nekrasov, A.A., Ivanov, V.F., and Vannikov, A.V., Abstracts of Papers, 50th ISE Meet., September 5–10, 1999, Pavia (Italy), Symp. 3b, abs. no. 484.
Nekrasov, A.A., Ivanov, V.F., and Vannikov, A.V., J. Electroanal. Chem., 2000, vol. 482, p. 11.
Nekrasov, A.A., Ivanov, V.F., and Vannikov, A.V., Elektrokhimiya, 2000, vol. 36, p. 1001.
Nekrasov, A.A., Ivanov, V.F., and Vannikov, A.V., Electrochim. Acta, 2001, vol. 46, p. 4051.
Nekrasov, A.A., Ivanov, V.F., and Vannikov, A.V., Electrochim. Acta, 2001, vol. 46, p. 3301.
Tanaka, J., Mashita, N., Mizoguchi, K., and Kume, K., Synth. Met., 1989, vol. 29, p. 175.
Genies, E.M., Lapkowski, M., and Tsintavis, C., New J. Chem., 1988, vol. 12, p. 181.
Neudeck, A., Petr, A., and Dunsch, L., Synth. Met., 1999, vol. 107, p. 143.
Zheng, W., Min, Y., MacDiarmid, A.G., Angelopoulos, M., Liao, Y.H., and Epstein, A.J., Synth. Met., 1997, vol. 84, p. 63.
Bernard, M.C., Bich, V.T., deTorresi, S.C., and Hugot-Le Goff, A., Synth. Met., 1997, vol. 84, p. 785.
Magon, C.J., Desouza, R.R., Costa, A.J., Vidoto, E.A., Faria, R.M., and Nascimento, O.R., J. Chem. Phys., 2000, vol. 112, p. 2958.
Zotti, G., Schiavon, G., and Zecchin, S., Synth. Met., 1995, vol. 72, p. 275.
Semenikhin, O.A., Ovsyannikova, E.V., Ehrenburg, M.R., Alpatova, N.M., and Kazarinov, V.E., J. Electroanal. Chem., 2000, vol. 494, p. 1.
Ivanov, V.F., Kucherenko, Yu.A., and Vannikov, A.V., Elektrokhimiya, 1993, vol. 29, p. 1146.
Ivanov, V.F. and Grishina, A.D., Izv. Akad. Nauk SSSR, 1977, p. 1873.
Vorotyntsev, M.A. and Heinze, J., Electrochim. Acta, 2001, vol. 46, p. 3309.
Abd-Elwahed, A. and Holze, R., Synth. Met., 2002, vol. 131, p. 61.
Ivanov, V.F., Kucherenko, Yu.A., Nekrasov, A.A., and Vannikov, A.V., Bull. Electrochem., 1992, vol. 8, p. 278.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nekrasov, A.A., Ivanov, V.F., Gribkova, O.L. et al. On the Role Played by Dimers of Radical Cations in the Process of Electrochemical Oxidation–Reduction of Polyaniline: The Data that Were Obtained Using the Method of Cyclic Voltabsorptometry in the Presence of Counteranions of a Diverse Nature. Russian Journal of Electrochemistry 40, 249–258 (2004). https://doi.org/10.1023/B:RUEL.0000019661.08569.28
Issue Date:
DOI: https://doi.org/10.1023/B:RUEL.0000019661.08569.28