Abstract
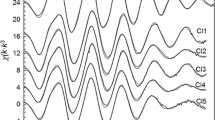
The literature data on the equilibrium constants of formation of HgCl– 3 solv and HgCl2– 4 solv from HgCl2 solv and Cl– solv in aqueous perchlorate–chloride solutions were generalized. The individual electronic absorption spectrum of the trichloro complex of mercury(II) was obtained for the first time, and the effect of the ionic strength on the spectra of di-, tri-, and tetrachloro complexes of mercury(II) was considered. The previously developed procedures for the resolution of absorption spectra into individual bands were found to be suitable for interpretation of spectral changes experimentally observed for solutions as the result of stepwise complexation. The parameters of the absorption bands in the spectra of chloro complexes of mercury(II) were determined.
Similar content being viewed by others
REFERENCES
Sillen, L.G., Acta Chem. Scand., 1949, vol. 3,no. 4, p. 539.
Marcus, Y., Acta Chem. Scand., 1957, vol. 11,no. 4, p. 599.
Panthaleon van Eck, C.L., Thesis, Leyden, 1958.
Eliezer, I. and Marcus, Y., J. Inorg. Nucl. Chem., 1963, vol. 25,no. 11, p. 1465.
Latysheva, V.A., Nechaev, K.P., and Savel'eva, Z.A., Khimiya i termodinamika rastvorov (Chemistry and Thermodynamics of Solutions), Leningrad: Len. Gos. Univ., 1964, p. 105.
Arnek, R., Arkiv Kemi, 1965, vol. 24,no. 6, p. 531.
Giavatta, L. and Grimaldi, M., J. Inorg. Nucl. Chem., 1968, vol. 30,no. 1, p. 197.
Sjoberg, S., Acta Chem. Scand., 1968, vol. 31,no. 3, p. 705.
Vasil'ev, V.P., Kozlovskii, E.V., and Mokeev, A.A., Zh. Neorg. Khim., 1982, vol. 27,no. 6, p. 1466.
Griffiths, T.R. and Anderson, R.A., J. Chem. Soc., Faraday Trans. 1, 1984, vol. 80,no. 10, p. 2361.
Spiro, T.G. and Hume, D.N., J. Am. Chem. Soc., 1961, vol. 83,no. 21, p. 4305.
Belevantsev, V.I. and Mironov, I.V., Zh. Neorg. Khim., 1982, vol. 27,no. 6, p. 1393.
Belevantsev, V.I. and Badmaeva, Zh.O., Zh. Neorg. Khim., 1980, vol. 20,no. 12, p. 3216.
Belevantsev, V.I. and Peshchevitskii, B.I., Issledovanie slozhnykh ravnovesii v rastvore (Study of Complex Equilibria in Solutions), Novosibirsk: Nauka, 1978, p. 9.
Belevantsev, V.I., Postanovka i opisanie issledovanii slozhnykh ravnovesii v rastvorakh (Statement and Description of the Study of Complex Equilibria in Solutions), Novosibirsk: Novosibirsk Gos. Univ., 1987, pp. 19, 71.
Belevantsev, V.I., Sokolovskaya, I.P., and Malkova, V.I., Sib. Zh., 1992, no. 2, p. 39.
Malkova, V.I. and Belevantsev, V.I., Trudy Sibirskoi konferentsii po prikladnoi i industrial'noi matematike (Proc. Siberian Conf. on Applied and Industrial Mathematics), Novosibirsk: IM, 1995, vol. 2, p. 146.
Makotchenko, E.V., Malkova, V.I., and Belevantsev, V.I., Koord. Khim., 1999, vol. 25,no. 4, p. 302.
Karyakin, Yu.V. and Angelov, I.I., Chistye khimicheskie veshchestva (Pure Chemical Substances), Moscow: Khimiya, 1974, p. 287.
Vasil'ev, V.P., Termodinamicheskie svoistva rastvorov elektrolitov (Thermodynamic Properties of Electrolyte Solutions), Moscow: Vysshaya Shkola, 1982, p. 320.
Belevantsev, V.I., Mironov, I.V., and Malkova, V.I., Zh. Neorg. Khim., 1983, vol. 28,no. 3, p. 547.
Belevantsev, V.I., Gushchina, L.V., and Obolenskii, A.A., Gidrotermal'noe nizkotemperaturnoe rudoobrazovanie i metasomotoz (Hydrothermal Low-Temperature Ore-Formation and Metasomatism), Novosibirsk: Nauka, 1982, p. 3.
Belevantsev, V.I., Zh. Fiz. Khim., 2002, vol. 76,no. 4, p. 608.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Belevantsev, V.I., Malkova, V.I., Gushchina, L.V. et al. Chloro Complexes of Mercury(II) in Aqueous Perchlorate Media: Equilibrium and Electronic Absorption Spectra. Russian Journal of Coordination Chemistry 30, 465–472 (2004). https://doi.org/10.1023/B:RUCO.0000034786.68915.69
Issue Date:
DOI: https://doi.org/10.1023/B:RUCO.0000034786.68915.69