Abstract
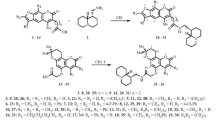
The reactions of 5,6,7,8-tetrafluoro-4-hydroxycoumarin derivatives with o-phenylenediamine occur with pyrone heterocycle cleavage and formation of substituted benzodiazepin-2-ones. 5,6,7,8-Tetrafluoro-4-hydroxycoumarin affords 4-(3,4,5,6-tetrafluoro-2-hydroxyphenyl)-2,3-dihydro-1H-1,5-benzodiazepin-2-one, 3-acetimidoyl-5,6,7,8-tetrafluoro-4-hydroxycoumarin produces 3-(3,4,5,6-tetrafluoro-2-hydroxybenzoyl)-4-methyl-1,2-dihydro-1H-1,5-benzodiazepin-2-one, and 3-acetyl-5,6,7,8-tetrafluoro-4-hydroxycoumarin yields both these heterocycles.
Similar content being viewed by others
References
G. A. Kuznetsova, Prirodnye kumariny i furokumariny [Natural Coumarins and Furocoumarins], Nauka, Leningrad, 1967, 248 pp. (in Russian).
Comprehensive Organic Chemistry, Eds S. D. Barton and W. D. Ollis, Pergamon Press, New York, 1979.
V. I. Saloutin, Z. E. Skryabina, I. T. Bazil´, and S. P. Kisil´, J. Fluorine Chem., 1999, 94, 83.
I. T. Bazyl´, S. P. Kisil´, A. E. Sharapko, Ya. V. Burgart, V. I. Saloutin, and O. N. Chupakhin, Zh. Org. Khim., 1998, 34, 394 [Russ. J. Org. Chem., 1998, 34 (Engl. Transl.)].
P. B. Terent´ev and A. P. Stankyavichus, Mass-spektrometriya biologicheski aktivnykh azotistykh osnovanii [Mass Spectrometry of Biologically Active Nitrogen Bases], Mokslas, Vil´nyus, 1987, 280 pp. (in Russian).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Burgart, Y.V., Shcherbakov, K.V., Saloutin, V.I. et al. 5,6,7,8-Tetrafluoro-4-hydroxycoumarin derivatives in reactions with o-phenylenediamine. Russian Chemical Bulletin 53, 1237–1239 (2004). https://doi.org/10.1023/B:RUCB.0000042279.90135.73
Issue Date:
DOI: https://doi.org/10.1023/B:RUCB.0000042279.90135.73