Abstract
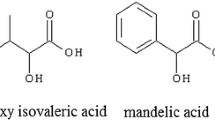
New calix[4]arenes, di- and tetrasubstituted at the lower rim, with different functional groups were synthesized. They were studied as carriers of a series of dicarboxylic and α-hydroxycarboxylic acids through a liquid impregnated membrane. The calix[4]arenes under study are capable of molecular recognition of oxalic acid in the series of structurally similar dicarboxylic and α-hydroxycarboxylic acids. The regularities found make it possible to change purposefully the receptor ability of 1,3-disubstituted calix[4]arenes by variation of the nature of substituents.
Similar content being viewed by others
References
V. Elliot and D. Elliot, Biokhimiya i molekulyarnaya biologiya [Biochemistry and Molecular Biology], MAIK Nauka/Interperiodika, Moscow, 2002, 444 pp. (in Russian).
J. H. Harley, T. D. James, and C. J. Ward, J. Chem. Soc., Perkin Trans. 1, 2000, 3155.
R. J. Fitzmaurice, G. M. Kyne, D. Douheret, and J. D. Kilburn, J. Chem. Soc., Perkin Trans. 1, 2002, 841.
T. H. Webb and C. S. Wilcox, Chem. Soc. Rev., 1993, 383.
T. Araki and H. Tsukube, Liquid Membranes: Chemical Application, CRC Press, Inc. Boca Raton, Florida, 1990, 213 p.
Z. Asfari, V. Bohmer, J. Harrowfield, J. Vicens, and M. Saadioui, Calixarenes 2001, Kluwer Academic Press, Dordrecht, 2001, 683 p.
I. S. Antipin, I. I. Stoikov, A. A. Khrustalev, and A. I. Konovalov, Izv. Akad. Nauk, Ser. Khim., 2001, 2038 [Russ. Chem. Bull., Int. Ed., 2001, 50, 1697].
N. J. Wolf, E. M. Georgiev, A. T. Yordanov, B. R. Whittlesey, H. F. Koch, and D. M. Roundhill, Polyhedron, 1999, 18, 885.a
F. Arnaud-Neu, E. M. Collins, M. Deasy, G. Ferguson, S. J. Harris, B. Kaitner, A. J. Lough, M. A. McKervey, E. Marques, B. L. Ruhl, M. J. Schwing-Weill, and E. M. Seward, J. Am. Chem. Soc., 1989, 111, 8681.
M. A. McKervey and E. M. Seward, J. Org. Chem., 1986, 51, 3583.
H. Akdas, G. Mislin, E. Graf, M. W. Hosseini, A. De Cian, and J. Fischer, Tetrahedron Lett., 1999, 40, 2116.
S. Yu. Ivakhno, A. V. Afanas´ev, and G. A. Yagodin, Itogi nauki i tekhniki, Neorganicheskaya khimiya [Results of Science and Technology, Inorganic Chemistry], VINITI AN SSSR, 1984, 13, 3 (in Russian).
E. S. Stern and C. J. Timmons, Gillam and Stern´s Introduction to Electronic Absorption Spectroscopy in Organic Chemistry, Edward Arnold (Publishers) Ltd, London, 1970, 295 pp.
S. Goswami, K. Ghosh, and R. Mukherjee, Tetrahedron, 2001, 57, 4987.
H. M. Colquhoun, E. P. Goodings, J. M. Maud, J. F. Stoddart, J. B. Wolstenholme, and D. J. Williams, J. Chem. Soc., Perkin Trans. 2, 1985, 607.
Qi-Yu Zheng, C.-F. Chen, and Z.-T. Huang, Tetrahedron, 1997, 53, 10345.
A. Casnati, Gazz. Chim. Ital., 1997, 127, 637.
C. D. Gutsche, B. Dhawan, J. A. Levine, Kwang Hyun No, and L. J. Bauer, Tetrahedron, 1982, 39, 409.
C. D. Gutsche, Calixarenes, Royal Society of Chemistry, Cambridge, 1989, 162.
I. I. Stoikov, O. A. Omran, S. E. Solovieva, Sh. K. Latypov, K. M. Enikeev, A. T. Gubaidullin, I. S. Antipin, and A. I. Konovalov, Tetrahedron, 2003, 59, 1469.
M. D. Joesten and L. J. Schaad, Hydrogen Bonding, Marcel Dekker, Inc., New York, 1974, 553 pp.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Stoikov, I.I., Gafiullina, L.I., Ibragimova, D.S. et al. Synthetic receptors based on calix[4]arene functionalized at the lower rim in molecular recognition of dicarboxylic, α-hydroxycarboxylic, and α-amino acids. Russian Chemical Bulletin 53, 1172–1180 (2004). https://doi.org/10.1023/B:RUCB.0000042270.26330.cb
Issue Date:
DOI: https://doi.org/10.1023/B:RUCB.0000042270.26330.cb