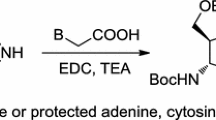
Abstract
The coupling of 5-acetoxy-1,1-dimethoxypent-2-ene with cytosine and thymine trimethylsilyl derivatives, as well as the reaction of 5-acetoxy-1-bromopent-2-ene with adenine sodium salt, yielded acyclic analogues of the corresponding nucleosides containing 5′-acetoxy groups. They were deprotected with a saturated methanolic solution of ammonia to the target analogues of nucleosides, which were characterized with 1H NMR, IR, and UV spectra.
Similar content being viewed by others
REFERENCES
Tsytovich, A.V., Shamshin, D.V., Burkovskii, V.B., and Shvets, V.I., Bioorg.Khim., 1995, vol. 21, pp. 874–880.
Tsytovich, A.V., Kochetkova, M.V., Kuznetsova, E.V., Mitsner, B.I., and Shvets, V.I., Bioorg.Khim., 1991, vol.17, pp. 1086–1093.
Mitsner, B.I., Kochetkova, M.V., Filippov, D.V., Tsytovich, A.V., and Dyatkina, N.B., Mol.Biol. (Moscow), 1993, vol. 27, pp. 174–184.
Malekin, S.I., Kruglyak, Yu.L., Khromova, N.Yu., Aksenova, M.Yu., Sokolov, V.P., Kisin, A.V., Novozhilkova, T.I., Popova, S.G., Kurochkin, V.K., Miller, G.G., and Pokidysheva, L.N., Bioorg.Khim., 1997, vol. 23, pp.648–654.
Florent'ev, V.L., Yavorskii, A.E., Kochetkova, S.V., Smirnov, I.P., Shchavel'eva, I.L., Tsylevich, T.L., and Gottikh, B.P., Bioorg.Khim., 1987, vol. 13, pp. 1000–1001.
Florent'ev, V.L., Yavorskii, A.E., and Reshot'ko, L.N., Khim.–Farm.Zh., 1988, vol. 22, pp. 833–836.
Florent'ev, V.L., Tsylevich, T.L., and Kochetkova, S.V., Bioorg.Khim., 1987, vol. 13, pp. 1240–1244.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vasilenko, I.A., Shamshin, D.V., Tsytovich, A.V. et al. Acyclic Nucleoside Analogues: III. A Synthesis of New 2′,3′-Dideoxy-2′,3′-Didehydronucleoside Analogues. Russian Journal of Bioorganic Chemistry 30, 547–552 (2004). https://doi.org/10.1023/B:RUBI.0000049771.66752.74
Issue Date:
DOI: https://doi.org/10.1023/B:RUBI.0000049771.66752.74