Abstract
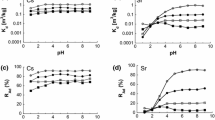
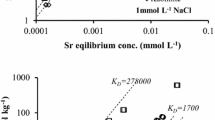
Joint sorption of Sr2 + and Fe3 + ions on blue Cambrian clay was studied. A relationship between the isotherm of ideal ion exchange and the Langmuir sorption isotherm was revealed. The apparent constant and Gibbs energy of exchange of Sr2 + and Fe3 + ions on blue Cambrian clay were calculated.
Similar content being viewed by others
REFERENCES
Chirkst, D.E., Litvinova, T.E., Cheremisina, O.V., and Streletskaya, M.I., Radiokhimiya, 2001, vol. 43, no. 5, pp. 475–478.
Analiticheskaya khimiya strontsiya (Analyutical Chemistry of Strontium), Moscow: Akad. Nauk SSSR, 1971.
Chirkst, D.E., Litvinova, T.E., Cheremisina, O.V., et al., Zh. Prikl. Khim., 2003, vol. 76, no. 6, pp. 922–925.
Chirkst, D.E., Krasotkin, I.S., Litvinova, T.E., et al., Zh. Prikl. Khim ., 2003, vol. 46, no. 4, pp. 687_ 689.
Analiticheskaya khimiya strontsiya (Analyutical Chemistry of Strontium), Moscow: Akad. Nauk SSSR, 1971.
Vasil'ev, V.P., Termodinamicheskie svoistva rastvorov elektrolitov (Thermodynamic Properties of Electrolyte Solutions), Moscow: Vysshaya Shkola, 1982.
Kokotov, Yu.A. and Pasechnik, V.A., Ravnovesie i kinetika ionnogo obmena(Equilibrium and Kinetics of Ion Exchange), Leningrad: Khimiya, 1970.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chirkst, D.E., Litvinova, T.E., Cheremisina, O.V. et al. Exchange Isotherm of Strontium(II) and Iron(III) Ions on Clay. Russian Journal of Applied Chemistry 77, 576–578 (2004). https://doi.org/10.1023/B:RJAC.0000038670.98156.51
Issue Date:
DOI: https://doi.org/10.1023/B:RJAC.0000038670.98156.51