Abstract
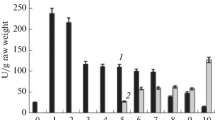
Photorespiratory enzyme serine:glyoxylate aminotransferase (SGAT, EC 2.6.1.45) was purified from green parts of seedlings of two Gramminae species with different photosynthetic pathways, maize (Zea mays L., C4 species) and wheat (Triticum aestivum L., C3 species). The preparation from wheat was homogeneous as judged by SDS-PAGE with silver staining for proteins; however, the same method revealed approximately 9% contamination in a highly purified maize preparation. Molecular masses of SGAT from maize and wheat were estimated by SDS-PAGE to be 44.1 and 44.6 kDa, respectively. C4 enzyme exhibited a specific activity in homogenates that was seven times lower than wheat, and this was associated with lower K m values for all substrates examined as well as a more than two times lower turnover number k cat with serine and glyoxylate as a pair of substrates. In contrast, the ratio of the turnover number to K m(Ser)(k cat/K m(Ser)) for C4 aminotransferase proved to be about two times higher than for C3 aminotransferase. The sensitivity of two enzymes to some inhibitors, especially aminooxyacetate, was different and they also differed with respect to thermal stability and pH optimum – the maize enzyme required 0.6 unit higher pH (8.6) for maximal activity and was more heat-resistant.
Similar content being viewed by others
References
Bauwe H and Kolukisaoglu LI (2003) Genetic manipulation of glycine decarboxylation. J Exp Bot 54: 1523–1535
Blum H, Bayer H and Gross HJ (1987) Improved silver staining of plant proteins, RNA and DNA in polyacrylamide gels. Electrophoresis 8: 93–99
Bradford MM (1976) A rapid and sensitive method for the quanti cation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248–254
Carp AJ and Smith JK (1974) Serine-glyoxylate aminotrans-ferase from kidney bean (Phaseolus vulgaris). Biochim Biophys Acta 370: 96–101
Crespo HM, Frean CF Cresswell CF and Tew J (1979) The occurrence of both C3 and C4 photosynthetic characteristics in single Zea mays plant. Planta 147: 257–263
Devi MT, Rajagopalan AV, Raghavendra AS (1996) Purification and properties of glycolate oxidase from plants with di. erent photosynthetic pathways: distinctness of C4 enzyme from that of a C3 species and a C3–C4 intermediate. Photosynth Res 47: 231–238
Doonan S (1996) Making and changing buffers. In: Doonan S (ed) Protein Puri cation Protocols, pp 103–113. Humana Press, Totowa, New Jersey
Dong LY, Masuda T, Kawamura T, Hata S and Izu K (1998) Cloning, expression and characterization of a root-form phosphoenolpyruvate carboxylase from Zea mays: comparison with the C4-form enzyme. Plant Cell Physiol 39: 865–873
Drincovich MF, Casati P and Andreo CS (2001) NADP-malic enzyme involved in di. erent metabolic pathways. FEBS Lett 490: 1–6
Foyer Ch H, Vanacker H, Gomez LD and Harbinson J (2002) Regulation of photosynthesis and antioxidant metabolism in maize leaves at optimal and chilling temperature: review. Plant Physiol Biochem 40: 659–668
Givan CV (1980) Aminotransferases in higher plants. In: Stumpf PK and Conn EE (eds) Biochemistry of Plants, pp 329–357. Academic Press, New York
Hath MD and Mau SL (1972) Activity, location and role of aspartate aminotransferase isoenzymes in leaves with C4 pathway photosynthesis. Arch Biochem Biophys 156: 195–206
Hondred D, McC Hunter J, Keith R, Titus DE and Becker WM (1985) Isolation of serine: glyoxylate aminotransferase from cucumber cotyledons. Plant Physiol 79: 95–102
Igamberdiev AU and Kleczkowski LA (1997) Glyoxylate metabolism during photorespiration-a cytosol connection. In: Pessarakli M (ed) Handbook of Photosynthesis, pp 269–279. Marcel Dekker, New York
Igarashi D, Miwa T, Seki M, Kobayashi M, Kato T, Tabata S, Shinozaki K and Ohsumi Ch (2003) Identification of photorespiratory glutamate: glyoxylate aminotransferase (GGAT) gene in Arabidopsis. Plant J 33: 975–987
Ireland RJ and Joy K (1983) Purification and properties of an asparagine aminotransferase from Pisum sativum leaves. Arch Biochem Biophys 223: 291–296
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685
Leegood RC (2002) C4 photosynthesis: principles of CO2 concentration and prospects for its introduction into C3 plants. J Exp Bot 53: 581–590
Leegood RC, Lea PJ, Adcook MD and Hausler R (1995) The regulation and control of photorespiration. J Exp Bot 46: 1397–1411
Liepman AH, and Olsen LJ (2001) Peroxisomal alanine: glyoxylate aminotransferase (AGT1) is a photorespiratory enzyme with multiple substrates in Arabidopsis thaliana. Plant J 25: 487–498
Liepman AH and Olsen LJ (2003) Alanine aminotransferase homologs catalyze the glutamate: glyoxylate aminotransferase reaction in peroxisomes of Arabidopsis. Plant Physiol 131: 215–227
Miyao M (2003) Molecular evolution and genetic engineering of C4 photosynthetic enzymes. J Exp Bot 54: 179–189
Noguchi T and Hayashi S (1980) Peroxisomal localization and properties of tryptophan aminotransferase in plant leaves. J Biol Chem 255: 2267–2269
Ohnishi J, Yamazaki M and Kanai R (1983) Differentiation of photorespiratory activity between mesophyl and bundle sheath cells of C4 plants II. Peroxisomes of Panicum miliaceum L. Plant Cell Physiol 26: 797–803
Orzechowski S, Socha-Hanc J and Paszkowski A (1999) Alanine aminotransferase and glycine aminotransferase from maize (Zea mays L.) leaves. Acta Biochim Polon 46: 447–457
Osmond CB, Badger M, Maxwell K, Bjorkman KO and Leegood RC (1997) Too many photons: photorespiration, photoinhibition and photooxidation. Trends Plant Sci 2: 119–121
Paszkowski A (1991) Some properties of serine: glyoxylate aminotransferase from rye seedlings (Secale cereale L.). Acta Biochim Polon 38: 437–448
Paszkowski A and Niedzielska A (1989) Glutamate: glyoxylate aminotransferase from the seedlings of rye (Secale cereale L.). Acta Biochim Polon 36: 17–29
Paszkowski and Niedzielska A (1990) Serine: glyoxylate aminotransferase from the seedlings of rye (Secale cereale L.). Acta Biochim Polon 37: 277–282
Reumann S (2000) The structural properties of plant peroxisomes and their metabolic signi cance. Biol Chem 381: 639–648
Smith JK (1985) Aminotransferases utilizing glyoxylate. In: Christen P and Metzler DE (eds) Transaminases, pp 390–396. John Wiley and Sons, New York
Voet D and Voet JG (1995) Biochemistry. John Wiley and Sons, New York
Walmsley A (1996) The effect of temperature on enzyme catalysed reactions In: Engel PC (ed) Enzymology Labfax, pp 190–198. Bios Scientific, Oxford
Ward DE, Kengen SWM, van der Oost J and de Vos WM (2000) Purification and characterization of the alanine aminotransferase from the hypertermophilic archeon Pyrococus furiosus and its role in alanine production. J Bacteriol 182: 2559–2566
Westhoff P, Svensson P, Ernst K, Blasing O, Burscheidt J and Stockhaus J (1997) Molecular evolution of C4 phosphoenolphyruvate carboxylase in the genus Flaveria. Aust J Plant Physiol 24: 429–436
Wilson K (1994) Protein and enzyme techniques. In: Wilson K and Walker J (eds) Practical Biochemistry, pp 162–226. Cambridge University Press, Cambridge, Massachusetts
Wojciechowski ChL, Cardia JP and Kantrowitz ER (2002) Alkaline phosphatase from the hypertermophilic bacterium T. maritima requires cobalt for activity. Protein Sci 11: 903–911
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Truszkiewicz, W., Paszkowski, A. Serine:Glyoxylate Aminotransferases from Maize and Wheat Leaves: Purification and Properties. Photosynthesis Research 82, 35–47 (2004). https://doi.org/10.1023/B:PRES.0000040435.35784.6b
Issue Date:
DOI: https://doi.org/10.1023/B:PRES.0000040435.35784.6b