Abstract
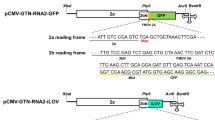
Abutilon mosaic virus (AbMV) and bean dwarf mosaic virus (BDMV) are two phylogenetically related bipartite begomoviruses. While AbMV is restricted to phloem, BDMV spreads to non-phloem tissues. Cell-to-cell and long-distance movement of AbMV and BDMV were investigated after replacing the coat protein (CP) gene with the reporter gene encoding the green fluorescence protein (GFP). The DNA-A and DNA-B genomic components of AbMV and BDMV, and their pseudorecombinants (PR), were delivered to bean (Phaseolus vulgaris) seedlings and detached leaves with DNA-coated microprojectiles. Virus-associated fluorescence was observed with the confocal microscope. Delivery of AbMV and BDMV GFP reporters showed that the epidermal tissue was the main recipient of the viral DNA; the DNA-A of the two viruses was unable to move out of the recipient cells. AbMV DNA-A co-inoculated with AbMV DNA-B did not move from cell to cell in the epidermis and did not reach the phloem. However, co-inoculation of AbMV DNA-A with BDMV DNA-B resulted in PR cell-to-cell movement out of the epidermis and long-distance movement in the phloem. In contrast, BDMV DNA-A moved from cell to cell and over a long distance when co-inoculated with either its own DNA-B or with the DNA-B of AbMV. Thus, the DNA-B of the non-phloem-limited BDMV overcame the phloem limitation of AbMV. In the reciprocal case, the DNA-B of the phloem-limited AbMV did not confine the non-phloem limited BDMV to the phloem. Hence, we assume that the DNA-A component of BDMV includes determinants involved in the movement pattern of the virus in addition to the DNA-B-encoded BC1 and BV1 which have previously been shown to be involved in virus movement. The results also confirm that the CP is not necessary for virus movement; however, replacing the CP of AbMV and BDMV with GFP resulted in a decrease in symptom severity. DNA-B was involved in symptom severity; the B component of BDMV produced symptoms more severe than those induced by that of AbMV, whether in wild-type PRs or in PRs with CP-GFP replacement. It is interesting to note that when the GFP gene under the control of the CaMV 35S promoter (35S-GFP) was delivered to the bean tissue, with or without the DNA-B component of BDMV, GFP was expressed but did not move from cell to cell. However, when the 35S-GFP was delivered together with BDMV DNA-A and DNA-B, GFP showed cell-to-cell movement in the epidermis but was restricted to these cells. Hence, infection of cells with a functional bipartite begomovirus may facilitate cell-to-cell movement of macromolecules.
Similar content being viewed by others
References
Ber, R., Navot, N., Zamir, D., Antignus, Y., Cohen, S. and Czosnek, H. 1990. Infection of tomato by the tomato yellow leaf curl virus: susceptibility to infection, symptom development, and accumulation of viral DNA. Arch. Virol. 112: 169–180.
Brown, J.K. 2001. The molecular epidemiology of begomoviruses. In: J.A. Khan and J. Dykstra (Eds.) Trends in Plant Virology, Haworth Press, New York, pp. 279–316.
Dellaporta, S.L., Wood, J. and Hicks, J.B. 1983. A plant DNA minipreparation: version II. Plant Mol. Biol. Rep. 4: 19–21.
Evans, D. and Jeske, H. 1993. DNA-B facilitates, but is not essential for the spread of Abutilon mosaic virus in agroinoculated Nicotiana benthamiana. Virology 194: 752–757.
Fauquet, C.M., Maxwell, D.P., Gronenborn, B., and Stanley, J. 2000. Revised proposal for naming geminiviruses. Arch. Virol. 145: 1743–1761.
Frischmuth, T., Zimmat, G. and Jeske, H. 1990. The nucleotide sequence of Abutilon mosaic virus reveals prokaryotic as well as eukaryotic features. Virology 178: 461–468.
Gallie, D.R., Sleat, D.E., Watts, J.W., Turner, P.C. and Wilson, T.M. 1987. The 5′-leader sequence of tobacco mosaic virus RNA enhances the expression of foreign gene transcripts in vitro and in vivo. Nucl. Acids Res. 15: 3257–3273.
Gilbertson, R.L., Faria, J.C., Hanson, S.F., Morales, F.J., Ahlquist, P., Maxwell, D.P. and Russell, D.R. 1991. Cloning of the complete DNA genomes of four bean-infecting geminiviruses and determining their infectivity by electric discharge particle acceleration. Phytopathology 81: 980–985.
Gilbertson, R.L., Hidayat, S.H., Paplomatas, E.J., Rojas, M.R., Hou, Y.M and Maxwell, D.P. 1993. Pseudorecombination between infectious cloned DNA components of tomato mottle and bean dwarf mosaic geminiviruses. J. Gen. Virol. 74: 23–31.
Goodman, R.M. 1977. Single-stranded DNA genome in a whiteflytransmitted plant virus. Virology 83: 171–179.
Groning, B.R., Frischmuth, T. and Jeske, H. 1990. Replicative form DNA of abutilon mosaic virus is present in plastids. Mol. Gen. Genet. 220: 485–488.
Hanley-Bowdoin, L., Settlage, S.B., Orozco, B.M., Nagar, S. and Robertson, D. 1999. Geminiviruses: models for plant DNA replication, transcription, and cell cycle regulation. Crit. Rev. Plant Sci. 18: 71–106.
Harrison, B.D. 1985. Advances in geminiviruses research. Annu. Rev. Phytopath. 23: 55–82.
Harrison, B.D., Barker, H., Bock, K.R., Guthrie, E.J., Meredith, G. and Atkinson, M. 1977. Plant viruses with circular singlestranded DNA. Nature 270: 760–762.
Höfer, P., Engel, M., Jeske, H. and Frischmuth, T. 1997. Host range limitation of a pseudorecombinant virus produced by two distinct bipartite geminiviruses. Mol. Plant-Microbe Interact. 10: 1019–1022.
Hidayat, S.H., Gilbertson, R.L., Hanson, S.F., Morales, F.J., Ahlquist, P., Russell, D.R. and Maxwell, D.P. 1993. Complete nucleotide sequences of the infectious cloned DNAs of bean dwarf mosaic geminivirus. Phytopathology 83: 181–187.
Höhnle, M., Höfer, P., Bedford, I.D., Briddon, R.W., Markham, P.G. and Frischmuth, T. 2001. Exchange of three amino acids in the coat protein results in efficient whitefly transmission of a nontransmissible Abutilon mosaic virus isolate. Virology 290: 164–171.
Horns, T. and Jeske, H. 1991. Localization of Abutilon mosaic virus (AbMV) DNA within leaf tissue by in situ hybridization. Virology 181: 580–588.
Hou, Y.M. and Gilbertson, R.J. 1996. Increased pathogenicity in a pseudorecombinant bipartite geminivirus correlates with intermolecular recombination. J. Virol. 70: 5430–5436.
Hou, Y.M., Paplomatas, E.J. and Gilbertson, R.J. 1998. Host adaptation and replication properties of two bipartite geminiviruses and their pseudorecombinants. Mol. Plant-Microbe Interact. 11: 208–217.
Imlau, A., Truernit, E. and Sauer, N. 1999. Cell-to-cell and longdistance trafficking of the green fluorescent protein in the phloem and symplastic unloading of the protein into sink tissues. Plant Cell 11: 309–322.
Moffat, A. 1999. Geminiviruses emerge as serious crop threat. Science 286: 1835.
Morales, F., Niessen, A., Ramirez, B. and Castaòo, M. 1990. Isolation and partial characterization of a geminivirus causing bean dwarf mosaic. Phytopathology 80: 96–101.
Morra, M.R. and Petty, T.D. 2000. Tissue specificity of geminivirus infection is genetically determined. Plant Cell 12: 2259–2270.
Oparka, K.J., Boevink, P. and Santa Cruz, S. 1996. Studying the movement of plant viruses using green fluorescent protein. Trends Plant Sci. 1: 412–418.
Oparka, K.J. and Roberts, A.G. 1999. Simple, but not branched, plasmodesmata allow the nonspecific trafficking of proteins in developing tobacco leaves. Cell 97: 743–754.
Rojas, M.R., Jian, H., Salati, R., Xoconostle-Cázares, B., Sudarshana, M.R., Lucas, W.L. and Gilbertson, R.L. 2001. Functional analysis of proteins involved in movement of the monopartite begomovirus, tomato yellow leaf curl virus. Virology 291: 110–125.
Sudarshana, M.R., Wang, H.L., Lucas, W.J. and Gilbertson, R.L. 1998. Dynamics of bean dwarf mosaic geminivirus cell-to-cell and long-distance movement in Phaseolus vulgaris revealed, using the green fluorescent protein. Mol. Plant-Microbe Interact. 11: 277–291.
Sunter, G. and Bisaro, D.M. 1997. Regulation of a Geminivirus coat protein promoter by AL2 protein (TrAP): evidence for activation and derepression mechanisms. Virology 232: 269–280.
van Regenmortel, M.H.V., Fauquet, C.M., Bishop, D.H.L., Carstens, E.B., Estes, M.K., Lemon, S.M., Maniloff, J., Mayo, M.A., McGeoch, D.J., Pringle, C.R. and Wickner, R.B. 2000. Virus Taxonomy: The Classification and Nomenclature of Viruses. Seventh Report of the International Committee on Taxonomy of Viruses. Academic Press, San Diego, CA, 1167 pp.
Wang, H.L., Gilbertson, R.L. and Lucas W.J. 1996. Spatial and temporal distribution of bean dwarf geminivirus in Phaseolus vulgaris and Nicotiana benthamiana. Phytopathology. 86: 1204–1214.
Wang, H.L., Sudarshana, M.R., Gilbertson, R.L. and Lucas, W.J. 1999. Analysis of cell-to-cell and long-distance movement of a Bean dwarf mosaic geminivirus-green fluorescent protein reporter in host and nonhost species: identification of sites of resistance. Mol. Plant-Microbe Interact. 12: 345–355.
Wege, C., Gotthardt, R.D., Frischmuth, T. and Jeske, H. 2000. Ful-filling Koch's postulates for Abutilon mosaic virus. Arch. Virol. 145: 2217–2225.
Zhang, W., Olson, N.H., Baker, T.S., Faulkner, L., Agbandje-McKenna, M., Boulton, M.I., Davis, J.W. and McKenna, R. 2001. Structure of the maize streak virus geminate particle. Virology 279: 471–477.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Levy, A., Czosnek, H. The DNA-B of the non-phloem-limited bean dwarf mosaic virus (BDMV) is able to move the phloem-limited Abutilon mosaic virus (AbMV) out of the phloem, but DNA-B of AbMV is unable to confine BDMV to the phloem. Plant Mol Biol 53, 789–803 (2003). https://doi.org/10.1023/B:PLAN.0000023662.25756.43
Issue Date:
DOI: https://doi.org/10.1023/B:PLAN.0000023662.25756.43