Abstract
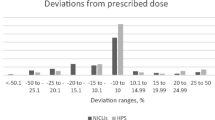
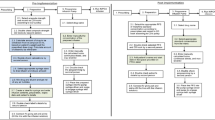
Objective: To describe current practices for i.v. drug admixture preparation, to identify potential improvements for the enhancement of patient safety.Design: A survey was conducted in a University hospital in Paris. Nurse practices were explored through the evaluation of five i.v. drug delivery systems: reconstituted freeze-dried drugs administered by syringe or i.v. bags, reconstituted drugs from vials administered by syringe or i.v. bag, i.v. bags Ready to Use (RtU).Participants: i.v. drug preparation practices were documented by a representative sample of nurses in the following departments: intensive care, emergency, abdominal surgery, cardiology, infectious diseases, hepatology.Main outcome measures: Data were collected regarding: existence of written procedures for preparations, sources of information, labelling, methods of preparation and calculation of doses, nurse satisfaction regarding safety and ease of use of the different i.v. systems.Results: A total of 299 questionnaires were completed and 100 nurses from the chosen wards were surveyed. The study highlighted a lack of procedure (71–85%) and a lack of labelling (37%). Conclusion: This survey highlighted areas for improvement in the preparation of i.v. drugs. It may contribute to raising awareness among nurses and physicians about the risks of medication errors. This survey also helped the pharmacy department in supporting the development of pharmaceutical procedures, the development of satellite pharmacy, the set up of training sessions for i.v. preparation and the switch toward ready to use packages when these are available.Current practices
Similar content being viewed by others
References
Kohn LT, Corrigan M, Donaldson MS, editors. For the Committee on Quality Health Care in America, Institute of Medicine. To Err is Human: Building a Safer Health System. Washington, DC: National Academy Press, 2000.
Akers MJ. Current problems and innovations in intravenous drug delivery. Am J Health Syst Pharm 1987; 44: 2528–30.
Bates DW, Cousins DD, Flynn E. et al. Consensus development conference statement on the safety of intravenous drug delivery systems: balancing safety and cost. Am J Health Syst Pharm 2000; 35(2): 150–5.
Lellouch J, Lazar P. Méthodes statistiques en expérimentation biologique. Médecine-Sciences Flammarion-Collection Statistique en Biologie et en Médecine. Paris: Flammarion, 1999.
Tissot E, Cornette C, Demoly P. et al. Medication errors at the administration stage in an intensive care unit. Intensive Care Med 1999; 25: 353–9.
Flynn EA, Pearson RE, Barker KN. Observational study of accuracy in compounding i.v. admixtures at five hospitals. Am J Health Syst Pharm 1997; 54: 904–12.
Limat S, Drouhin JP, Demesmay K et al. Incidence and risk factors of preparation errors in a centralized cytotoxic preparation unit. Pharm World Sci 2001; 23(3): 102–6.
Hoppe-Tischy T, Noe-Schwenn S, Wahlig A, Taxis K. Presentation of the study by the pharmacy of the Heidelberg Uniclinic. Medication errors with serious sequalae. Pflege Aktuell 2002; 56(9): 469–71.
Mansfield A, Cousins D. An observational study in four hospitals. 6th EAHP Congress March 2001, Amsterdam.
Loeb AJ, Fishman DA, Kochis TR. Premixed intravenous admixtures: A critical challenge for hospital pharmacy. Am J Hosp Pharm 1983; 40: 1041–3.
Cohen M. Drug product characteristics that faster drug-use system errors. Am J Health Syst Pharm 1995; 52: 395–9.
Cohen M, Anderson R, Antillio R et al. Preventing medication errors in cancer chemotherapy. Am J Health Syst Pharm 1996; 53: 737–46.
Lee HE. Premixed intravenous admixtures: A positive development for hospital pharmacy. Am J Hosp Pharm 1983; 40: 1043–4.
Wall DS, Noe LL, Abel SR et al. A resource use comparison of Monovial® with traditional methods of preparing extemporaneous small volume intravenous infusions. Hosp Pharm 1997; 12: 1647–56.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tilleul, P., Mons, B., Schmitt, C. et al. Intravenous drug preparation practices: a survey in a French university hospital. Pharm World Sci 25, 276–279 (2003). https://doi.org/10.1023/B:PHAR.0000006522.84493.4e
Issue Date:
DOI: https://doi.org/10.1023/B:PHAR.0000006522.84493.4e