Abstract
Purpose. We examined the effects of dose and dosing protocol on the pharmacodynamics of in vivo nitroglycerin (NTG) tolerance in conscious rats. Mechanism-based pharmcokinetic/pharmacodynamic (PK/PD) models were tested for their ability to describe the observed data.
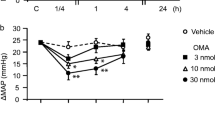
Methods. Rats were infused with 1, 3, or 10 μg/min of NTG or vehicle for 10 h. Peak mean arterial pressure (MAP) response to an hourly 30 μg i.v. NTG challenge dose (CD) was measured before, during, and at 12 and 24 h after infusion. In separate experiments, the MAP effects of repeated bolus doses of NTG were compared to those after a continuous infusion, both at a total dose of 510 μg NTG.
Results. NTG tolerance was indicated by a decrease in peak MAP response to the CD, relative to the preinfusion peak MAP response. Tolerance toward the MAP effects of bolus CD was observed within 1 h of 10 μg/min of NTG infusion (26.8 ± 2.8% vs. 10.6 ± 0.4% for 0 and 1 h, respectively, p < 0.001), and the rate and extent of tolerance development increased with the infusion dose. No apparent MAP tolerance was observed when NTG was given as multiple bolus doses whereas significant MAP tolerance was observed when this dose was infused continuously. PK/PD modeling indicated that a cofactor/enzyme depletion mechanism could adequately describe the composite data.
Conclusions. Our data showed that in vivo nitrate tolerance was dose- and dosing protocol-dependent. The pharmacodynamics of tolerance development are consistent with depletion of either critical enzymes or cofactors that are necessary to induce vasodilation.
Similar content being viewed by others
REFERENCES
H. L. Fung and J. A. Bauer. Mechanisms of nitrate tolerance. Cardiovasc. Drugs Ther. 8:489-499 (1994).
D. D. Stewart. Remarkable tolerance to nitroglycerin. Polyclinic 6:43(1888).
H. DeMots and S. P. Glasser. Intermittent transdermal nitroglycerin therapy in the treatment of chronic stable angina. J. Am. Coll. Cardiol. 13:786-795 (1989).
J. O. Parker and H. L. Fung. Transdermal nitroglycerin in angina pectoris. Am. J. Cardiol. 54:471-476 (1984).
K. Sakai and O. Kuromaru. Nitrate tolerance: comparison of nicorandil, isosorbide dinitrate, and nitroglycerin in anesthetized dogs. J. Cardiovasc. Pharmacol. 10:S17-S24 (1987).
J. A. Bauer and H. L. Fung. Effects of chronic glyceryl trinitrate on left ventricular hemodynamics in a rat model of congestive heart failure: demonstration of a simple animal model for the study of in vivo nitrate tolerance. Cardiovasc. Res. 24:198-203 (1990).
E. A. Kowaluk and H. L. Fung. Dissociation of nitrovasodilator-induced relaxation from cyclic GMP levels during in vitro nitrate tolerance. Eur. J. Pharmacol. 176:91-95 (1990).
C. M. Newman, J. B. Warren, G. W. Taylor, A. R. Boobis, and D. S. Davies. Rapid tolerance to the hypotensive effects of glyceryl trinitrate in the rat: prevention by N-acetyl-L-but not N-acetyl-D-cysteine. Br. J. Pharmacol. 99:825-829 (1990).
T. Munzel, H. Sayegh, B. A. Freeman, M. M. Tarpey, and D. G. Harrison. Evidence for enhanced vascular superoxide anion production in nitrate tolerance. A novel mechanism underlying tolerance and cross-tolerance. J. Clin. Invest. 95:187-194 (1995).
I. S. De la Lande, I. Stafford, and J. D. Horowitz. Effects of guanylyl cyclase and protein kinase G inhibitors on vasodilatation in non-tolerant and tolerant bovine coronary arteries. Eur. J. Pharmacol. 370:39-46 (1999).
B. P. Booth, S. Jacob, J. A. Bauer, and H. L. Fung. Sustained antiplatelet properties of nitroglycerin during hemodynamic tolerance in rats. J. Cardiovasc. Pharmacol. 28:432-438 (1996).
H. L. Fung, S. C. Sutton, and A. Kamiya. Blood vessel uptake and metabolism of organic nitrates in the rat. J. Pharmacol. Exp. Ther. 228:334-341 (1984).
J. A. Bauer and H. L. Fung. Pharmacodynamic models of nitroglycerin-induced hemodynamic tolerance in experimental heart failure. Pharm. Res. 11:816-823 (1994).
S. J. Chung and H. L. Fung. Relationship between nitroglycerin-induced vascular relaxation and nitric oxide production. Probes with inhibitors and tolerance development. Biochem. Pharmacol. 45:157-163 (1993).
P. Needleman, B. Jakschik, and E. M. Johnson, Jr. Sulfhydryl requirement for relaxation of vascular smooth muscle. J. Pharmacol. Exp. Ther. 187:324-331 (1973).
P. Needleman and E. M. Johnson, Jr. Mechanism of tolerance development to organic nitrates. J. Pharmacol. Exp. Ther. 184:709-715 (1973).
S. J. Chung, S. Chong, P. Seth, C. Y. Jung, and H. L. Fung. Conversion of nitroglycerin to nitric oxide in microsomes of the bovine coronary artery smooth muscle is not primarily mediated by glutathione-S-transferases. J. Pharmacol. Exp. Ther. 260:652-659 (1992).
W. I. Lee and H. L. Fung. Mechanism-based partial inactivation of glutathione S-transferases by nitroglycerin: tyrosine nitration vs sulfhydryl oxidation. Nitric Oxide 8:103-110 (2003).
H. L. Fung and R. Poliszczuk. Nitrosothiol and nitrate tolerance. Z. Kardiol. 75:25-27 (1986).
N. L. Dayneka, V. Garg, and W. J. Jusko. Comparison of four basic models of indirect pharmacodynamic responses. J. Pharmacokinet. Biopharm. 21:457-478 (1993).
J. E. Shaffer, B. J. Han, W. H. Chern, and F. W. Lee. Lack of tolerance to a 24-hour infusion of S-nitroso N-acetylpenicillamine (SNAP) in conscious rabbits. J. Pharmacol. Exp. Ther. 260:286-293 (1992).
D. Zimrin, N. Reichek, K. T. Bogin, G. Aurigemma, P. Douglas, B. Berko, and H. L. Fung. Antianginal effects of intravenous nitroglycerin over 24 hours. Circulation 77:1376-1384 (1988).
J. C. Cowan, J. P. Bourke, D. S. Reid, and D. G. Julian. Prevention of tolerance to nitroglycerin patches by overnight removal. Am. J. Cardiol. 60:271-275 (1987).
H. L. Fung. Clinical pharmacology of organic nitrates. Am. J. Cardiol. 72:9C–13C; discussion 14C–15C (1993).
R. Morris and A. Munkarah. Alternate dosing schedules for topotecan in the treatment of recurrent ovarian cancer. Oncologist 7(Suppl 5):29-35 (2002).
C. H. Kleinbloesem, P. van Brummelen, and D. D. Breimer. Nifedipine. Relationship between pharmacokinetics and pharmacodynamics. Clin. Pharmacokinet. 12:12-29 (1987).
P. S. Yap and H. L. Fung. Pharmacokinetics of nitroglycerin in rats. J. Pharm. Sci. 67:584-586 (1978).
K. E. Torfgard, J. Ahlner, K. L. Axelsson, B. Norlander, and A. Bertler. Tissue disposition of glyceryl trinitrate, 1,2-glyceryl dinitrate, and 1,3-glyceryl dinitrate in tolerant and nontolerant rats. Drug Metab. Dispos. 20:553-558 (1992).
K. Hasegawa, T. Taniguchi, K. Takakura, Y. Goto, and I. Muramatsu. Possible involvement of nitroglycerin converting step in nitroglycerin tolerance. Life Sci. 64:2199-2206 (1999).
P. R. Sage, I. S. de la Lande, I. Stafford, C. L. Bennett, G. Phillipov, J. Stubberfield, and J. D. Horowitz. Nitroglycerin tolerance in human vessels: evidence for impaired nitroglycerin bioconversion. Circulation 102:2810-2815 (2000).
E. Q. Wang, W. I. Lee, D. Brazeau, and H. L. Fung. cDNA microarray analysis of vascular gene expression after nitric oxide donor infusions in rats: implications for nitrate tolerance mechanisms. AAPS PharmSci 4:E10(2002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, E.Q., Balthasar, J.P. & Fung, HL. Pharmacodynamics of in Vivo Nitroglycerin Tolerance in Normal Conscious Rats: Effects of Dose and Dosing Protocol. Pharm Res 21, 114–120 (2004). https://doi.org/10.1023/B:PHAM.0000012158.80991.7e
Issue Date:
DOI: https://doi.org/10.1023/B:PHAM.0000012158.80991.7e