Abstract
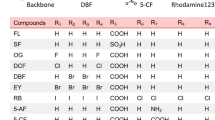
Purpose. To synthesize fluorescent analogues of hPept1 substrates, FITC-Val-OCH3, Lys-FITC-OH, and Lys-FITC-OCH3, and to characterize their hPept1 transporter-mediated uptake.
Methods. FITC analogues of amino acids were synthesized using established synthetic procedures, and the extent of their [3H]Gly-Sar uptake inhibition in HeLa/hPept1 cells was determined. The uptake of Lys-FITC-OCH3 was evaluated in HeLa, HeLa/hPept1, and Caco-2 cells in the presence and absence of Gly-Sar using a fluorescence microscopy-based assay. The uptake and transport of the Lys-FITC analogues were also determined in Caco-2 cells using HPLC assays.
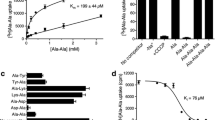
Results. In HeLa/hPept1 cells, [3H]Gly-Sar uptake was significantly inhibited by Lys-FITC-OCH3 (74%) but not by FITC-Val-OCH3 (22%). The uptake of Lys-FITC-OCH3 (100 μM) was approximately 10-fold higher in HeLa/hPept1 cells. Also, Lys-FITC-OCH3 (100 μM) uptake in HeLa/hPept1 and Caco-2 cells was reduced by 77% and 80%, respectively, in the presence of 1 mM Gly-Sar. Dipeptides and cephalexin significantly reduced Lys-FITC-OCH3 uptake in Caco-2 cells. The apical permeability of Lys-FITC-OCH3 (1.5 × 106 cm/s) in Caco-2 cells was significantly lowered in the presence of Gly-Sar. Fluorescence micrographs revealed that this analogue was localized in the cytoplasm and in the nucleus.
Conclusions. The combined results indicate that Lys-FITC-OCH3 is recognized and transported by hPept1 in HeLa/hPept1 and by peptide transporters in Caco-2 cells. The results also suggest that Lys-FITC-OCH3 might be a useful fluorescent substrate for rapid assessment of peptide transporter activity in cells of interest.
Similar content being viewed by others
References
W. Kramer, C. Dechent, F. Girbig, U. Gutjahr, and H. Neubauer. Intestinal uptake of dipeptides and beta-lactam antibiotics. I. The intestinal uptake system for dipeptides and beta-lactam antibiotics is not part of a brush border membrane peptidase. Biochim. Biophys. Acta 1030:41-49 (1990).
K. Inui, Y. Tomita, T. Katsura, T. Okano, M. Takano, and R. Hori. H+ coupled active transport of bestatin via the dipeptide transport system in rabbit intestinal brush-border membranes. J. Pharmacol. Exp. Ther. 260:482-486 (1992).
M. Hu and G. L. Amidon. Passive and carrier-mediated intestinal absorption components of captopril. J. Pharm. Sci. 77:1007-1011 (1988).
M. A. Jacobson, P. de Miranda, D. M. Cederberg, T. Burnette, E. Cobb, H. R. Brodie, and J. Mills. Human pharmacokinetics and tolerance of oral ganciclovir. Antimicrob. Agents Chemother. 31:1251-1254 (1987).
P. de Miranda and M. R. Blum. Pharmacokinetics of acyclovir after intravenous and oral administration. J. Antimicrob. Chemother. 12(ppl B):29-37 (1983).
S. Weller, M. R. Blum, M. Doucette, T. Burnette, D. M. Cederberg, P. de Miranda, and M. L. Smiley. Pharmacokinetics of the acyclovir pro-drug valaciclovir after escalating single-and multiple-dose administration to normal volunteers. Clin. Pharmacol. Ther. 54:595-605 (1993).
D. Jung and A. Dorr. Single-dose pharmacokinetics of valganciclovir in HIV-and CMV-seropositive subjects. J. Clin. Pharmacol. 39:800-804 (1999).
H. K. Han, D. M. Oh, and G. L. Amidon. Cellular uptake mechanism of amino acid ester prodrugs in Caco-2/hPEPT1 cells overexpressing a human peptide transporter. Pharm. Res. 15:1382-1386 (1998).
M. Sugawara, W. Huang, Y. J. Fei, F. H. Leibach, V. Ganapathy, and M. E. Ganapathy. Transport of valganciclovir, a ganciclovir prodrug, via peptide transporters PEPT1 and PEPT2. J. Pharm. Sci. 89:781-789 (2000).
D. E. Gonzalez, K. M. Covitz, W. Sadee, and R. J. Mrsny. An oligopeptide transporter is expressed at high levels in the pancreatic carcinoma cell lines AsPc-1 and Capan-2. Cancer Res. 58:519-525 (1998).
C. J. Whitaker, S. H. Battah, M. J. Forsyth, C. Edwards, R. W. Boyle, and E. K. Matthews. Photosensitization of pancreatic tumour cells by delta-aminolaevulinic acid esters. Anticancer Drug Des. 15:161-170 (2000).
D. A. Groneberg, F. Doring, P. R. Eynott, A. Fischer, and H. Daniel. Intestinal peptide transport: ex vivo uptake studies and localization of peptide carrier PEPT1. Am. J. Physiol. Gastrointest. Liver Physiol. 281:G697-G704 (2001).
H. Abe, M. Satoh, S. Miyauchi, S. Shuto, A. Matsuda, and N. Kamo. Conjugation of dipeptide to fluorescent dyes enhances its affinity for a dipeptide transporter (PEPT1) in human intestinal Caco-2 cells. Bioconjug. Chem. 10:24-31 (1999).
C. P. Hsu, J. M. Hilfinger, E. Walter, H. P. Merkle, B. J. Roessler, and G. L. Amidon. Overexpression of human intestinal oligopeptide transporter in mammalian cells via adenoviral transduction. Pharm. Res. 15:1376-1381 (1998).
H.K. Han, R. L. A. de Vrueh, J. K. Rhie, K.M. Y. Covitz, P. L. Smith, C.P. Lee, D.M. Oh, W. Sadee, and G. L. Amidon. 5′-Amino acid esters of antiviral nucleosides, acyclovir, and AZT are absorbed by the intestinal PEPT1 peptide. Pharm. Res. 15:1154-1189 (1998).
E. Walter, T. Kissel, and G. L. Amidon. The intestinal peptide carrier: A potential transport system for small peptide derived drugs. Adv. Drug Deliv. Rev. 20:33-58 (1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Landowski, C.P., Han, Hk., Lee, KD. et al. A Fluorescent hPept1 Transporter Substrate for Uptake Screening. Pharm Res 20, 1738–1745 (2003). https://doi.org/10.1023/B:PHAM.0000003369.64891.51
Issue Date:
DOI: https://doi.org/10.1023/B:PHAM.0000003369.64891.51