Abstract
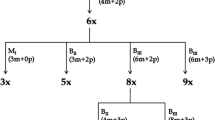
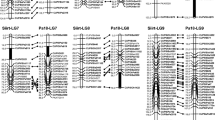
Tetraploid Paspalum notatum (bahiagrass) is a valuable forage grass with aposporous apomictic reproduction. In a previous study, we showed that apospory in bahiagrass is under the control of a single dominant gene with a distorted segregation ratio. The objective of this work was to identify molecular markers linked to apospory in tetraploid P. notatum and establish a preliminary syntenic relationship with the genomic region associated with apospory in P. simplex. A F1 population of 290 individuals, segregating for apospory, was generated after crossing a completely sexual plant (Q4188) with a natural aposporous apomictic plant (Q4117). The whole progeny was classified as sexual or aposporous by embryo sacs analysis. A bulked segregant analysis was carried out to identify molecular markers co-segregating with apospory. Four hundred RAPD primers, 30 AFLP primers combinations and 85 RFLP clones were screened using DNA from both parental genotypes and aposporous and sexual bulks. Linkage analysis was performed with cytological and genetic information from the complete progeny. Cytoembryological analysis showed 219 sexual and 71 aposporous F1 individuals. Seven different molecular markers (2 RAPD, 4 AFLP and 1 RFLP) were found to be completely linked to apospory. The RFLP probe C1069, mapping to the telomeric region of the long arm of rice chromosome 12, was one of the molecular markers completely linked to apospory in P. notatum. This marker had been previously associated with apospory in P. simplex. A preliminary map of the chromosome region carrying the apospory locus was constructed.
Similar content being viewed by others
References
Asker S.E. and Jerling L. 1992. Apomixis in plants. CRC Press, Boca Raton, Florida, USA.
Barcaccia G.A., Mazzucato E., Albertini J., Zethof A., Gerats M., Pezzotti M. and Falcinelli M. 1998. Inheritance of parthenogenesis in Poa pratensis L.: auxin test and AFLP linkage analyses support monogenic control. Theor. Appl. Genet. 97: 74–82.
Burton G.W. 1948. The method of reproduction in common Bahia Grass, Paspalum notatum. J. Am. Soc. Agron. 40: 443–452.
Carman J.G. 1997. Asynchronous expression of duplicate genes in angiosperms may cause apomixis, bispory, tetraspory, and polyembryony. Biol. J. Linn. Soc. 61: 51–94.
Chase A. 1929. The North American species of Paspalum. Contr. U. S. Natl. Herb. 28: 1–310.
Demarly Y. 1979. The concept of linkat. Proceed. Conf. on Broadening Genet. Base of Crops, Wageningen, 1978. Pudoc, Wageningen, The Netherlands. pp. 257–265.
do Valle C.B., Glienke C. and Leguizamon G.O.C. 1994. Inheritance of apomixis in Brachiaria, a tropical forage grass. Apomixis Newsl. 7: 42–43.
Grimanelli D., Leblanc O., Espinosa E., Perotti E., González de León D. and Savidan Y. 1998a. Mapping diplosporous apomixis in tetraploid Tripsacum: one gene or several genes? Heredity 80: 33–39.
Grimanelli D., Leblanc O., Espinosa E., Perotti E., González De León D. and Savidan Y. 1998b. Non-Mendelian transmission of apomixis in maize-Tripsacum hybrids caused by a transmission ratio distortion. Heredity 80: 40–47.
Grossniklaus U., Koltunow A. and van Lookeren Campagne M. 1998. A bright future for apomixis. Trends Plant Sci. 3: 415–416.
Gustine D.L., Sherwood R.T. and Huff D.R. 1997. Apospory-linked molecular markers in buffelgrass. Crop Sci. 37: 947–951.
Hanna W.W. and Bashaw E.C. 1987. Apomixis: its identification and use in plant breeding. Crop Sci. 27: 1136–1139.
Jefferson R.A. 1993. Strategic development of apomixis as a general tool for agriculture. In: Wilson K.J. (ed.), Proceed. International Workshop on Apomixis in Rice, Jan. 13-15, 1992, CAMBIA, Changsha, China. Canberra, Australia. pp. 206–217.
Jefferson R.A. 1994. Apomixis: a social revolution for agriculture? The Monitor 19: 14–16.
Koltunow A.M. 1993. Apomixis: embryo sacs and embryos formed without meiosis or fertilization in ovules. Plant Cell 5: 1425–1437.
Koltunow A.M., Bicknell R.A. and Chaudhury A.M. 1995. Apomixis: Molecular strategies for the generation of genetically identical seeds without fertilization. Plant Physiol. 108: 1345–1352.
Leblanc O., Grimanelli D., González De León D. and Savidan Y. 1995. Detection of the apomictic mode of reproduction in maize-Tripsacum hybrids using maize RFLP markers. Theor. Appl. Genet. 90: 1198–1203.
Martínez E.J., Urbani M.H., Quarin C.L. and Ortiz J.P.A. 2001. Inheritance of apospory in bahiagrass, Paspalum notatum. Hereditas 135: 19–25.
Michelmore R.W., Paran I. and Kesselli V. 1991. Identification of markers linked to disease-resistance genes by bulked segregant analysis: A rapid method to detect markers in specific genomic regions by using segregating populations. Proc. Natl. Acad. Sci. USA. 88: 9828–9832.
Nagamura Y., Antonio B.A. and Sasaki T. 1997. Rice molecular genetic map using RFLPs and its applications. Plant Mol. Biol. 35: 79–87.
Naumova T.N. 1993. Apomixis in Angiosperms: nucellar and integumentary embryony. CRC Press, Boca Raton, Florida, USA.
Nogler G.A. 1984. Gametophytic apomixis. In: Johri B.M. (ed.), Embryology of angiosperms. Springer-Verlag, Berlin, pp. 475–518.
Noyes R.D. and Rieseberg L.H. 2000. Two independent loci control agamospermy (apomixis) in the triploid flowering plant Erigeron annuus. Genetics 155: 379–390.
Ortiz J.P.A., Pessino S.C., Leblanc O., Hayward M.D. and Quarin C.L. 1997. Genetic fingerprinting for determining the mode of reproduction in Paspalum notatum, a subtropical apomictic forage grass. Theor. Appl. Genet. 95: 850–856.
Ozias-Akins P., Lubbers E.L., Hanna W.W. and McNay J.W. 1993. Transmission of the apomictic mode of reproduction in Pennisetum: co-inheritance of the trait and molecular markers. Theor. Appl. Genet. 85: 632-638.
Ozias-Akins P., Roche D. and Hanna W.W. 1998. Tight clustering and hemizygosity of apomixis-linked molecular markers in Pennisetum squamulatum implies genetic control of apospory by a divergent locus that may have no allelic form in sexual genotypes. Proc. Natl. Acad. Sci. USA. 95: 5127–5132.
Pessino S.C., Ortiz J.P.A., Leblanc O., do Valle C.B. and Hayward M.D. 1997. Identification of a maize linkage group related to apomixis in Brachiaria. Theor. Appl. Genet. 94: 439–444.
Pessino S.C., Evans C., Ortiz J.P.A., Armstead I., do Valle C.B. and Hayward M.D. 1998. A genetic map of the apospory-region in Brachiaria hybrids: identification of two markers closely associated with the trait. Hereditas 128: 153–158.
Pessino S.C., Ortiz J.P.A., Hayward M.D. and Quarin C.L. 1999. The molecular genetics of gametophytic apomixis. Hereditas 130: 1–11.
Pupilli F., Lambobarda P., Cáceres M.E., Quarin C.L. and Arcioni S. 2001. The chromosome segment related to apomixis in Paspalum simplex is homoeologous to the telomeric region of the long arm of rice chromosome 12. Mol. Breed. 8: 53–61.
Quarin C.L. 1992. The nature of apomixis and its origin in panicoid grasses. Apomixis Newsl. 5: 8–15.
Richards A.J. 1986. Plant Breeding Systems. Allen & Unwin, London, UK.
Roche D., Cong P., Chen Z., Hanna W.W., Gustine D.L., Sherwood R.T. and Ozias-Akins P. 1999. An apospory-specific genomic region is conserved between buffelgrass (Cenchrus ciliaris L.) and Pennisetum squamulatum Fresen. Plant J. 19: 203–208.
Roche D., Chen Z., Hanna W.W. and Ozias-Akins P. 2001. Non-Mendelian transmission of an apospory-specific genomic region in a reciprocal cross between sexual pearl millet (Pennisetum glaucum) and an apomictic F1 (P. glaucum x P. squamulatum). Sex. Plant Reprod. 13: 217-223.
Sagahai-Maroof M.A., Soliman K.M., Jorgensen R.A. and Allard R.W. 1984. Ribosomal DNA spacer-length polymorphisms in barley: Mendelian inheritance, chromosomal location, and population dynamics. Proc. Natl. Acad. Sci. USA. 81: 8014–8018.
Savidan Y. 1982. Nature et hérédité de l'apomixie chez Panicum maximum Jacq. Travaux et Documents OSTROM, Paris. 153: 1-159.
Savidan Y. 2000. Apomixis: Genetics and breeding. In: Janick J. (ed.), Plant Breeding Reviews. John Wiley & Sons, Inc. 18: 13-86.
Sherwood R.T., Berg C.C. and Young B.A. 1994. Inheritance of apospory in Buffelgrass. Crop Sci. 34: 1490–1494.
Stam P. 1993. Construction of integrated genetic linkage maps by means of a new computer package: JoinMap. Plant J. 5: 739–744.
Stebbins G.L. 1950. Variation and evolution in plants. Columbia Univ Press, New York, USA.
van Deynze A.E., Sorrells M.E., Park W.D., Ayres N.M., Fu H., Cartinhour S.W., Paul E. and McCouch S.R. 1998. Anchor probes for comparative mapping in grass genera. Theor. Appl. Genet. 97: 356-369.
Vos P.R., Hogers R., Bleeker M., Reijans M., Van de Lee T., Hornes M., Frijters A., Pot J., Peleman J., Kuiper M. and Zabeau M. 1995. AFLP: a new technique for DNA fingerprinting. Nucleic Acids Res. 23: 4407-4414.
Wu K.K., Burnquist W., Sorrells M.E., Tew T.L., Moore P.H. and Tanksley S.D. 1992. The detection and estimation of linkage in polyploids using single-dose restriction fragments. Theor. Appl. Genet. 83: 294-300.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Martínez, E.J., Hopp, H.E., Stein, J. et al. Genetic characterization of apospory in tetraploid Paspalum notatum based on the identification of linked molecular markers. Molecular Breeding 12, 319–327 (2003). https://doi.org/10.1023/B:MOLB.0000006868.12568.32
Issue Date:
DOI: https://doi.org/10.1023/B:MOLB.0000006868.12568.32