Abstract
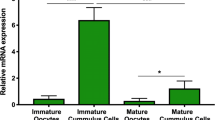
The endothelial type (NOS-3) of three isoforms of nitric oxide (NO) synthase occurs in porcine oocytes and granulosa cells, but the regulation of NO synthesis in oocytes remains unknown. The present study was designed to evaluate steroid control in the process of oocyte NO synthesis. Cumulus-oocyte complexes (COCs), obtained from small-sized antral follicles of immature porcine ovaries, were cultured in estrogen-deprived medium, and the effect of steroids or steroid-free porcine follicular fluids on the NO release from oocytes was investigated. Oocytes that were isolated from cultured COCs were incubated with 1 μM ionomycin. The NO metabolites were identified using a NO detector-high-pressure liquid chromatography system. Oocytes from COCs cultured with 10 nM 17β-estradiol (E2) released NO in response to ionomycin, whereas progesterone and testosterone had little effect on the synthesis of NO. An inhibitor of NOS suppressed the synthesis of NO. The maximal synthesis was observed after a 15 h-culture with E2. However, oocytes freshly obtained from antral follicles did not response to ionomycin, and the E2 action was suppressed by the addition of steroid-free follicular fluids. Analyses of RT-PCR and Western blotting showed that E2 did not increase NOS-3 expression. In addition, estrogen receptor β was detected in oocytes and cumulus cells, and estrogen receptor α was detected only in cumulus cells. These findings suggest that oocyte NOS-3 is promoted for the synthesis of NO by E2 without increases in NOS-3 expression, but the synthesis of NO is suppressed, at least in the oocytes of early antral follicles.
Similar content being viewed by others
References
Van Voorhis BJ, Moore K, Strijbos PJL, Nelson S, Baylis SA, Grzybicki D, Weiner CP: Expression and localization of inducible and endothelial nitric oxide synthase in the rat ovary. J Clin Invest 96: 2719-2726, 1995
Jablonka-Shariff A, Olson LM: Hormonal regulation of nitric oxide synthases and their cell-specific expression during follicular development in the rat ovary. Endocrinology 138: 460-468, 1997
Van Voorhis BJ, Dunn MS, Snyder GD, Weiner CP: Nitric oxide: An autocrine regulator of human granulosa-luteal cell steroidogenesis. Endocrinology 135: 1799-1806, 1994
Jablonka-Shariff A, Olson LM: The role of nitric oxide in oocyte meiotic maturation and ovulation: Meiotic abnormalities of endothelial nitric oxide synthase knock-out mouse oocytes. Endocrinology 139: 2944-2954, 1998
Basini G, Baratta M, Ponderato N, Bussolati S, Tamanini C: Is nitric oxide an autocrine modulator of bovine granulosa cell function? Reprod Fertil Dev 10: 471-478, 1998
Hattori M-A, Nishida N, Takesue K, Kato Y, Fujihara N: FSH suppression of nitric oxide synthesis in porcine oocytes. J Mol Endocrinol 24: 65-73, 2000
Nishida N, Takesue K, Hattori M-A, Kato Y, Wakabayashi K, Fujihara N: Modulatory action of nitric oxide on the expression of transcription factor gene, c-fos and c-jun, in developing porcine granulosa cells in vitro. J Reprod Dev 46: 167-175, 2000
Moncada S, Palmer PMJ, Higgs EA: Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol Rev 43: 109-142, 1991
Nathan C: Nitric oxide as a secretory product of mammalian cells. FASEB J 6: 3051-3064, 1992
Lander HM, Jacovina AT, Davis RJ, Tauras JM: Differential activation of mitogen-activated protein kinase by nitric oxide-treated species. J Biol Chem 271: 19705-19709, 1996
Olson LM, Jones-Burton CM, Jablonka-Shariff A: Nitric oxide decreases estradiol synthesis of rat luteinized ovarian cells: Possible role for nitric oxide in functional luteal regression. Endocrinology 137: 3531-3539, 1996
Snyder GD, Holmes RW, Batas JN, Van Voorhis BJ: Nitric oxide inhibits aromatase activity: Mechanisms of actions. J Steroid Biochem Mol Biol 58 63-69, 1996
Salvemini D, Misko TP, Masferrer JL, Seibert K, Currie MG, Needleman P: Nitric oxide activates cyclooxygenase enzymes. Proc Natl Acad Sci USA 90: 7240-7244, 1993
Corbett JA, Kwon G, Turk J, McDaniel ML: IL-1β induces the coexpression of both nitric oxide synthase and cyclooxygenase by islets of Langerhans: Activation of cyclooxygenase by nitric oxide. Biochemistry 32: 13767-13770, 1993
Hattori M-A, Takesue K, Kato Y, Fujihara N: Expression of endothelial nitric oxide synthase gene in the porcine oocyte and its possible function. Mol Cell Biochem 219: 121-126, 2001
Drazen DL, Klein SL, Burnett AL, Wallach EE, Crone JK, Huang PL, Nelson RJ: Reproductive function in female mice lacking the gene for endothelial nitric oxide synthase. Nitric Oxide 3: 366-374, 1999
Takesue K, Hattori M-A, Nishida N, Kato Y, Fujihara N: Expression of endothelial nitric oxide synthase gene in cultured porcine granulosa cells after FSH stimulation. J Mol Endocrinol 26: 259-265, 2001
Nathan C, Xie QW: Regulation of biosynthesis of nitric oxide. J Biol Chem 269: 13725-13728, 1994
Venkov CD, Rankin AB, Vaughan DE: Identification of authentic estrogen receptor in cultured endothelial cells. A potential mechanism for steroid hormone regulation of endothelial function. Circulation 94: 727-33, 1996
Iruela-Arispe ML, Rodriguez-Manzaneque JC, Abu-Jawdeh G: Endometrial endothelial cells express estrogen and progesterone receptors and exhibit a tissue specific response to angiogenic growth factors. Microcirculation 6: 127-140, 1999
Caulin-Glaser T, Garcia-Cardena G, Sarrel P, Sessa WC, Bender JR: 17β-Estradiol regulation of human endothelial cell basal nitric oxide release, independent of cytosolic Ca2+ mobilization. Cir Res 81: 885-892, 1997
Chen Z, Yuhanna IS, Galcheva-Gargova Z, Karas RH, Mendelsohn ME, Shaul PW: Estrogen receptor α mediates the nongenomic activation of endothelial nitric oxide synthase by estrogen. J Clin Invest 103: 401-406, 1999
McNeill AM, Kim N, Duckles SP, Krause DN, Kontos HA: Chronic estrogen treatment increases levels of endothelial nitric oxide synthase protein in rat cerebral microvessels. Stroke 30: 2186-2190, 1999
Slomczynska M, Duda M, Galas J: Estrogen receptor α and β expression in the porcine ovary. Folia Histochem Cytobiol 39: 137-138, 2001
Wu TC, Wang L, Wan YJ: Detection of estrogen receptor messenger ribonucleic acid in human oocytes and cumulus-oocyte complexes using reverse transvriptase-polymerase chain reaction. Fertil Steril 59: 54-59, 1993
Hiroi H, Momoeda M, Inoue S, Tsuchiya F, Matsumi H, Tsutsumi O, Muramatsu M, Taketani Y: Stage-specific expression of estrogen receptor subtypes and estrogen responsive finger protein in preimplantational mouse embryos. Endocrine J 46: 153-158, 1999
Brower PT, Schultz RM: Intercellular communication between granulosa cells and mouse oocytes: Existence and possible nutritional role during oocyte growth. Dev Biol 90: 144-153, 1982
Hyttel P, Greve T, Callesen H: Ultrastructural aspects of oocyte maturation and fertilization in cattle. J Reprod Fertil 38: 35-47, 1989
Vallance P, Leone A, Calver A, Collier J, Moncada S: Accumulation of an endogenous inhibitor of nitric oxide synthesis in chronic renal failure. Lancet 339: 572-575, 1992
Park KH, Rubin LE, Gross SS, Levi R: Nitric oxide is a mediator of hypoxic coronary vasodilation. Relation to adenosine and cyclooxygenase-derived metabolites. Circ Res 71: 992-1001, 1992
Ackland JF, Schwartz NB, Mayo KE, Dodson RE: Nonsteroidal signals originating in the gonads. Physiol Rev 72: 731-787, 1992
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hattori, Ma., Arai, M., Saruwatari, K. et al. Estrogen regulation of nitric oxide synthesis in the procine oocyte. Mol Cell Biochem 260, 13–19 (2004). https://doi.org/10.1023/B:MCBI.0000026046.32875.70
Issue Date:
DOI: https://doi.org/10.1023/B:MCBI.0000026046.32875.70