Abstract
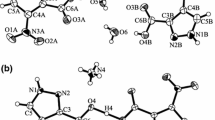
New solid complexes of a herbicide known as dicamba (3,6-dichloro-2-methoxybenzoic acid) with Pb(II), Cd(II), Cu(II) and Hg(II) of the general formula M(dicamba)2·xH2O (M=metal, x=0-2) and Zn2(OH)(dicamba)3·2H2O have been prepared and studied. The complexes have different crystal structures. The carboxylate groups in the lead, cadmium and copper complexes are bidentate, chelating, symmetrical, in Hg(dicamba)2·2H2O - unidentate, and in the zinc salt - bidentate, bridging, symmetrical. The anhydrous compounds decompose in three stages, except for the lead salt whose decomposition proceeds in four stages. The main gaseous decomposition products are CO2, CH3OH, HCl and H2O. Trace amounts of compounds containing an aromatic ring were also detected. The final solid decomposition products are oxychlorides of metals and CuO.
Similar content being viewed by others
References
L. Róaski, Vademecum of pesticides '97/98, Agra-Enviro Lab., Pozna 1998
H. Zhao, W. F. Jaynes and G. F. Vance, Chemosphere, 33 (1996) 2089
C. H. L. Kennard, B. Kerr, E. J. O'Reilly and G. Smith, Aust. J. Chem., 37 (1984) 1757
G. Smith, E. J. O'Reilly and C. H. L. Kennard, Aust. J. Chem., 36 (1983) 2175
J. Koby ecka, B. Ptaszyski, R. Rogaczewski and A. Turek, Thermochim. Acta, 407 (2003) 25.
J. Kobyecka and A. Turek, Ann. Pol. Chem. Soc., 2 (2003) 462
B. Ptaszyski and A. Zwoliska, J. Therm. Anal. Cal., 74 (2003) 237
B. Ptaszyski and A. Zwoliska, J. Therm. Anal. Cal., 75 (2004) 301
F. J. Welcher, The Analytical Uses of Ethylenediaminetetraacetic Acid, WNT, Warszawa1958
B. S. Manhas and A. K. Trikha, J. Indian Chem. Soc., 59 (1982) 315
G. B. Deacon and R. J. Phillips, Coord. Chem. Rev., 33 (1980) 227
Powder Diffraction File. International Center of Diffraction Date. ICPDS-ICDD 1601 Park Lane Swarthmore1990
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Turek, A., Kobyłecka, J. & Ptaszyński, B. Complexes of dicamba with cadmium(II), copper(II), mercury(II), lead(II) and zinc(II). Journal of Thermal Analysis and Calorimetry 78, 513–524 (2004). https://doi.org/10.1023/B:JTAN.0000046115.64116.95
Issue Date:
DOI: https://doi.org/10.1023/B:JTAN.0000046115.64116.95