Abstract
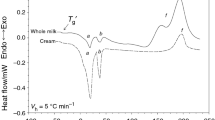
The effects of heat treatment on soymilk protein denaturation were studied by differential scanning calorimetry (DSC) and electrophoresis. Transition behavior of soymilk was studied by DSC. Three endotherms were found in DSC heating curves; the transition observed at around 70°C is attributed to the denaturation of 7S (b-conglycinin) and the transition at around 90°C is to 11S (glycinin). The denaturation temperature increased with the increasing soymilk protein content. The change of electrophoretic patterns after heat treatments indicated that soy proteins were dissociated into subunits, some of which coalesced. When the heating temperature is below their denaturation temperature, the protein fractions cannot completely be denatured even after heat exposure for extended periods of time.
Similar content being viewed by others
References
D. Fukushima, Food Sci. Technol. Res., 7 (2001) 8.
A. M. Hermansson, J. Texture Stud., 9 (1978) 33.
K. C. Kwok and K. Niranjan, Int. J. Food Sci. Tech., 30 (1995) 263.
J. de dios Alvarado, J. Food Proc. Eng., 14 (1991) 189.
K. A. Thorarinsdottir, S. Arason, M. Geirsdottir, S. G. Bogason and K. Kristbergsson, Food Chem., 77 (2002) 377.
B. I. Kurganov, A. E. Lyubarev, J. M. Sanchez-Ruiz and V. L. Shnyrov, Biophys. Chem., 69 (1997) 125.
S. F. Wang and D. M. Smith, Poultry Sci. Rev., 5 (1994) 145.
P. Relkin, Thermochim. Acta, 246 (1994) 371.
J. W. Donovan and R. A. Beardslee, J. Biol. Chem., 250 (1975) 1966.
M. Lu, B. Wang, Zh. Li, Y. Fei, L. Wei and Sh. Gao, J. Therm. Anal. Cal., 67 (2002) 689.
A. Raemy, J. Therm. Anal. Cal., 71 (2003) 273.
AACC, proved Methods of the AACC 10th edition, method 44 (2000) 110.
J. M. Berg, J. L. Tymoczko and L. Stryer, Biochemistry, W. H. Freeman and Company, New York, 2001, p. 84.
U. K. Laemmli, Nature, 227 (1970) 680.
V. Neuhoff, N. Arold, D. Taube and N. Ehrhardt, Electrophoresis, 9 (1988) 255.
C. Y. Ma and V. R. Harwalkar, Adv. Food and Nutr. Res., 35 (1991) 317.
E. A. S. Rosa and A. S. Rodrigues, HortScience, 36 (2001) 56.
M. M. FalcĂo-Rodrigues, M. MoldĂo-Martins and M. L. BeirĂo-da-Costa, Eur. Food Res. Technol., 215 (2002) 317.
F. Yamauchi, T. Yamagishi and S. Iwabuchi, Food Rev. Int., 7 (1991) 283.
S. Utsumi, A. B. Gidamis, J. Kanamori, I. J. Kang and M. Kito, J. Agric. Food Chem., 41 (1993) 687.
N. Kitabatake, M. Tahara and E. Doi, Agri. Biol. Chem., 54 (1990) 2205.
W. M. Jackson and J. F. Brandts, Biochemistry, 9 (1970) 2294.
S. D. Arntfiel and E. D. Murray, Can. Inst. Food Sci. Technol. J., 14 (1981) 289.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, H., Takenaka, M. & Isobe, S. DSC and electrophoretic studies on soymilk protein denaturation. Journal of Thermal Analysis and Calorimetry 75, 719–726 (2004). https://doi.org/10.1023/B:JTAN.0000027168.18317.78
Issue Date:
DOI: https://doi.org/10.1023/B:JTAN.0000027168.18317.78