Abstract
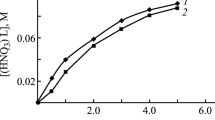
Solvent extraction of tetravalent thorium and trivalent europium ions from nitrate media into dichloromethane solution of triphenylphosphine oxide (TPPO) has been studied. The extractant was shown to be more efficient for europium than for thorium. A conventional log-log analysis reveals that the extraction of both metal nitrates takes place via the formation of the species with 1 : 2 metal to ligand ratio. Thermodynamic parameters i.e., ΔG°, ΔH° and ΔS° of the extraction process using 1,2-dichloroethane as diluent have been calculated based on the influence of the temperature on extraction equilibria in the range 293-313 K. While the extraction of europium is controlled by enthalpy changes, the extraction of thorium is an endothermic process and is driven by entropy changes. A comparison of these data with those obtained for the extraction of europium and thorium nitrates by two other related phosphorylated ligands, tri-n-octylphosphine oxide (TOPO) and diphenyl-N,N-dimethylcarbamoylmethylphosphine oxide (DФDMCMPO), indicates that DФDMCMPO coordinates presumably as a chelating ligand.
Similar content being viewed by others
References
B. Weaver, Solvent Extraction in the Separation of Rare Earths and Trivalent Actinides, in Ion Exchange and Solvent Extraction, Vol. 6, J. A. Marinsky and Y. Marcus (Eds), Marcel Dekker, New York, 1974.
E. P. Horwitz, R. Chiarizia, in: Separation Techniques in Nuclear Waste Management, N. A. Chipmenand C. M. Wai (Eds), CRC, FL, 1996.
W. W. Schulz, L. L. Burger, J. D. Navratil (Eds), Science and Technology of Tributylphosphate, Vols I-III, CRC, Boca Raton, FL., 1984-1990.
E. P. Horwitz, D. G. Kalina, A. C. Muscatello, Separ. Sci. Technol., 16 (1981) 403.
E. P. Horwitz, D. G. Kalina, L. Kaplan, G. W. Mason, H. Diamond, Separ. Sci. Technol., 17 (1982) 1261.
E. P. Horwitz, K. A. Martin, H. Diamond, L. Kaplan, Solvent Extr. Ion Exch., 4 (1986) 449
E. P. Horwitz, D. G. Kalina, H. Diamond, G. F. Vandegrift, Solvent Extr. Ion Exch., 3 (1985) 75.
D. G. Kalina, Solvent Extr. Ion Exch., 2 (1984) 381.
T. Nakamura, C. Miyake, Solvent Extr. Ion Exch., 12 (1994) 931.
T. Takeuchi, S. Tanaka, M. Yamawaki, Solvent Extr. Ion Exch., 12 (1994) 987.
M. W. Peters, E. J. Werner, M. J. Scott, J. Inorg. Chem., 41 (2002) 1707.
M. R. Yaftian, M. Burgard, C. Wieser, C. B. Dieleman, Solvent Extr. Ion Exch., 16 (1998) 1131.
M. R. Yaftian, L. Hassanzadeh, M. E. Eshraghi, D. Matt, Separ. Pur. Tech., 31 (2003) 261.
M. R. Yaftian, M. E. Eshraghi, L. Hassanzadeh, Iran. Chem. Chem. Eng., 22 (2003) 71.
Z. Marczenko, Spectrophotometric Determination of Elements, Wiley, New York, 1976.
F. Arnaud-Neu, V. Böhmer, J.-F. Dozol, C. Grüttner, R. A. Jakobi, D. Kraft, O. Mauprivez, H. Roquette, M.-J. Schwing-Weill, N. Simon, W. Vogt, J. Chem. Soc., Perkin Trans. II, (1996) 1175.
M. R. Yaftian, R. Taheri, D. Matt, Phosphorus, Sulfur, Silicon, 178 (2003) 1225.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yaftian, M.R., Taheri, R., Zamani, A.A. et al. Thermodynamics of the solvent extraction of thorium and europium nitrates by neutral phosphorylated ligands. Journal of Radioanalytical and Nuclear Chemistry 262, 455–459 (2004). https://doi.org/10.1023/B:JRNC.0000046777.74156.6f
Issue Date:
DOI: https://doi.org/10.1023/B:JRNC.0000046777.74156.6f