Abstract
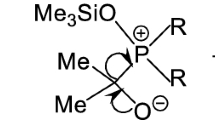
The structures and energies of the tautomeric forms of salicylphosphorous and phthalimidophosphorous acids have been calculated by the DFT method. The tautomeric forms are found to be in equilibrium. Both tautomeric forms of phthalimidophosphorous acid may have an intramolecular hydrogen bond.
Similar content being viewed by others
REFERENCES
É. E. Nifantiev, Hydrophosphoryl Chemistry [in Russian], Nauka, Moscow (1983).
V. M. Mamaev, A. V. Prisyajnyuk, D. N. Laikov, et al., Mendeleev Commun., 8 No.6, 240-241 (1999).
V. M. Mamaev, A. V. Prisyajnyuk, L. S. Logutenko, and Yu. V. Babin, ibid., 10 No.6, 221-222 (2001).
V. V. Ovchinnikov, S. V. Cherezov, R. A. Cherkasov, and A. N. Pudovik, Zh. Org. Khim., 54 No.5, 1021-1030 (1984).
S. A. Mamedov, F. Yu. Aliev, and O. V. Karaeva, ibid., 60 No.5, 1183-1184 (1990).
J. P. Perdew, K. Burke, and M. Ernzerhof, J. Chem. Phys., 105, 9982-9992 (1996).
D. N. Laikov, Chem. Phys. Lett., 281, 151-156 (1997).
D. N. Laikov, “An economical approach to DFT calculations of molecules and its application to complex chemical problems,” Physical and Mathematical Sciences Candidate' s Dissertation, Moscow (2000).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kalinov, S.M., Prisyazhnyuk, A.V. & Babin, Y.V. Theoretical Investigation of the Prototropic Diad Rearrangement of Salicylphosphorous and Phthalimidophosphorous Acids. Journal of Structural Chemistry 44, 874–876 (2003). https://doi.org/10.1023/B:JORY.0000029828.91403.bc
Issue Date:
DOI: https://doi.org/10.1023/B:JORY.0000029828.91403.bc