Abstract
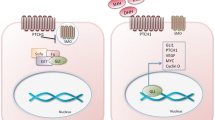
The hedgehog signal transduction network is a critical mediator of cell-cell communication during embryonic development. Evidence also suggests that properly regulated hedgehog network function is required in some adult organs for stem cell maintenance or renewal. Mutation, or misexpression, of network genes is implicated in the development of several different types of cancer, particularly that of skin, brain, lung, and pancreas. Recent studies in the mouse mammary gland have demonstrated roles for hedgehog network genes at virtually every phase of mammary gland development where it regulates such diverse processes as embryonic mammary gland induction, establishment of ductal histoarchitecture, and functional differentiation in lactation. Further, studies suggest a role for misregulated network function in the progression of breast cancer.
Similar content being viewed by others
REFERENCES
Cohen MM, Jr. The hedgehog signaling network. Am J Med Genet 2003;123A:5-28.
Ingham PW, McMahon AP. Hedgehog signaling in animal development: paradigms and principles. Genes Dev 2001;15:3059-87.
Bailey EC, Scott MP, Johnson RL. Hedgehog signaling in animal development and human disease. Ernst Schering Res FoundWorkshop 2000:211-35.
McMahonAP.More surprises in the Hedgehog signaling pathway. Cell 2000;100:185-8.
Murone M, Rosenthal A, de Sauvage FJ. Hedgehog signal transduction: from flies to vertebrates. Exp Cell Res 1999;253:25-33.
Ruiz i Altaba A. Gli proteins and Hedgehog signaling: development and cancer. Trends Genet 1999;15:418-25.
Lewis MT. Hedgehog signaling in mouse mammary gland development and neoplasia. J Mammary Gland Biol Neoplasia 2001;6:53-66.
Machold R, Hayashi S, Rutlin M, Muzumdar MD, Nery S, Corbin JG, et al. Sonic hedgehog is required for progenitor cell maintenance in telencephalic stem cell niches. Neuron 2003;39:937-50.
Watkins DN, Berman DM, Burkholder SG, Wang B, Beachy PA, Baylin SB. Hedgehog signalling within airway epithelial progenitors and in small-cell lung cancer. Nature 2003;422:313-7.
Oro AE, Higgins K. Hair cycle regulation of Hedgehog signal reception. Dev Biol 2003;255:238-48.
Ruiz i Altaba A, Sanchez P, Dahmane N. Gli and hedgehog in cancer: tumours, embryos and stem cells. Nat Rev Cancer 2002;2:361-72.
Wicking C, McGlinn E. The role of hedgehog signalling in tumorigenesis. Cancer Lett 2001;173:1-7.
Taipale J, Beachy PA. The Hedgehog and Wnt signalling pathways in cancer. Nature 2001;411:349-54.
Toftgard R. Hedgehog signalling in cancer. Cell Mol Life Sci 2000;57:1720-31.
Zeng X, Goetz JA, Suber LM, Scott WJ, Jr., Schreiner CM, Robbins DJ. A freely diffusible form of Sonic hedgehog mediates long-range signalling. Nature 2001;411:716-20.
Gritli-Linde A, Lewis P, McMahon AP, Linde A. The whereabouts of a morphogen: direct evidence for short-and graded long-range activity of hedgehog signaling peptides. Dev Biol 2001;236:364-86.
Burke R, Nellen D, Bellotto M, Hafen E, Senti KA, Dickson BJ, et al. Dispatched, a novel sterol-sensing domain protein dedicated to the release of cholesterol-modified hedgehog from signaling cells. Cell 1999;99:803-15.
Ma Y, Erkner A, Gong R, Yao S, Taipale J, Basler K, et al. Hedgehog-mediated patterning of the mammalian embryo requires transporter-like function of dispatched. Cell 2002;111:63-75.
Taipale J, Cooper MK, Maiti T, Beachy PA. Patched acts catalytically to suppress the activity of Smoothened. Nature 2002;418:892-7.
Johnson RL, Milenkovic L, Scott MP. In vivo functions of the patched protein: requirement of the C terminus for target gene inactivation but not Hedgehog sequestration. Mol Cell 2000;6:467-78.
Thibert C, Teillet MA, Lapointe F, Mazelin L, Le Douarin NM, Mehlen P. Inhibition of neuroepithelial patched-induced apoptosis by sonic hedgehog. Science 2003;301:843-6.
Guerrero I, Ruiz i Altaba A. Development. Longing for ligand: hedgehog, patched, and cell death. Science 2003;301:774-6.
Chao MV. Dependence receptors: what is the mechanism? Sci STKE 2003;2003:PE38.
Zako M, Dong J, Goldberger O, Bernfield M, Gallagher JT, Deakin JA. Syndecan-1 and-4 synthesized simultaneously by mouse mammary gland epithelial cells bear heparan sulfate chains that are apparently structurally indistinguishable. J Biol Chem 2003;278:13561-9.
Prince JM, Klinowska TC, Marshman E, Lowe ET, Mayer U, Miner J, et al. Cell-matrix interactions during development and apoptosis of the mouse mammary gland in vivo. Dev Dyn 2002;223:497-516.
Liu BY, Kim YC, Leatherberry V, Cowin P, Alexander CM. Mammary gland development requires syndecan-1 to create a beta-catenin/TCF-responsive mammary epithelial subpopulation. Oncogene 2003;22:9243-53.
Delehedde M, Lyon M, Sergeant N, Rahmoune H, Fernig DG. Proteoglycans: pericellular and cell surface multireceptors that integrate external stimuli in the mammary gland. J Mammary Gland Biol Neoplasia 2001;6:253-73.
Zcharia E, Metzger S, Chajek-Shaul T, Friedmann Y, Pappo O, Aviv A, et al. Molecular properties and involvement of heparanase in cancer progression and mammary gland morphogenesis. J Mammary Gland Biol Neoplasia 2001;6:311-22.
Kasai K, Takahashi M, Osumi N, Sinnarajah S, Takeo T, Ikeda H, et al. The G12 family of heterotrimeric G proteins and Rho GTPase mediate Sonic hedgehog signalling. Genes Cells 2004;9:49-58.
Ruel L, Rodriguez R, Gallet A, Lavenant-Staccini L, Therond PP. Stability and association of Smoothened, Costal2 and Fused with Cubitus interruptus are regulated by Hedgehog. Nat Cell Biol 2003;5:907-13.
Ogden SK, Ascano M, Jr., Stegman MA, Suber LM, Hooper JE, Robbins DJ. Identification of a functional interaction between the transmembrane protein Smoothened and the kinesin-related protein Costal2. Curr Biol 2003;13:1998-2003.
Lum L, Zhang C, Oh S, Mann RK, von Kessler DP, Taipale J, et al. Hedgehog signal transduction via Smoothened association with a cytoplasmic complex scaffolded by the atypical kinesin, Costal-2. Mol Cell 2003;12:1261-74.
Jia J, Tong C, Jiang J. Smoothened transduces Hedgehog signal by physically interacting with Costal2/Fused complex through its C-terminal tail. Genes Dev 2003;17:2709-20.
Sakanaka C, Sun TQ, Williams LT. New steps in the Wnt/betacatenin signal transduction pathway. Recent Prog Horm Res 2000;55:225-36.
Nusse R. Wnts and Hedgehogs: lipid-modified proteins and similarities in signaling mechanisms at the cell surface. Development 2003;130:5297-305.
Kalderon D. Similarities between the Hedgehog and Wnt signaling pathways. Trends Cell Biol 2002;12:523-31.
Ingram WJ, Wicking CA, Grimmond SM, Forrest AR, Wainwright BJ. Novel genes regulated by Sonic Hedgehog in pluripotent mesenchymal cells. Oncogene 2002;21:8196-205.
Bonifas JM, Pennypacker S, Chuang PT, McMahon AP, Williams M, Rosenthal A, et al. Activation of expression of hedgehog target genes in basal cell carcinomas. J Invest Dermatol 2001;116:739-42.
Yoon JW, Kita Y, Frank D, Majewski RR, Konicek BA, Nobrega MA, et al. Gene expression profiling leads to identification of GLI1 binding elements in target genes and a role for multiple downstream pathways in GLI1 induced cell transformation. J Biol Chem 2001.
Louro ID, Bailey EC, Li X, South LS, McKie-Bell PR, Yoder BK, et al. Comparative gene expression profile analysis ofGLI and c-MYC in an epithelial model of malignant transformation. Cancer Res 2002;62:5867-73.
Robinson GW, Karpf AB, Kratochwil K. Regulation of mammary gland development by tissue interaction. J Mammary Gland Biol Neoplasia 1999;4:9-19.
Pispa J, Thesleff I. Mechanisms of ectodermal organogenesis. Dev Biol 2003;262:195-205.
Daniel CWS, G.B. Developmental biology of the mammary gland. In Daniel MCNaCW, editor. The Mammary Gland. New York: Plenum; 1987.
Sakakura T. Mammary embryogenesis. In Daniel MCNaCW, editor. The Mammary Gland. New York: Plenum; 1987.
Veltmaat JM, Mailleux AA, Thiery JP, Bellusci S. Mouse embryonic mammogenesis as a model for the molecular regulation of pattern formation. Differentiation 2003;71:1-17.
Daniel CW, Smith GH. The mammary gland: a model for development. J Mammary Gland Biol Neoplasia 1999;4:3-8.
Veltmaat JM, Van Veelen W, Thiery JP, Bellusci S. Identifi-cation of the mammary line in mouse by Wnt10b expression. Dev Dyn 2004;229:349-56.
Kimata K, Sakakura T, Inaguma Y, Kato M, Nishizuka Y. Participation of two different mesenchymes in the developing mouse mammary gland: synthesis of basement membrane components by fat pad precursor cells. J Embryol Exp Morphol 1985;89:243-57.
Foley J, Dann P, Hong J, Cosgrove J, Dreyer B, Rimm D, et al. Parathyroid hormone-related protein maintains mammary epithelial fate and triggers nipple skin differentiation during embryonic breast development. Development 2001;128:513-25.
Sakakura T, Nishizuka Y, Dawe CJ. Mesenchyme-dependent morphogenesis and epithelium-specific cytodifferentiation in mouse mammary gland. Science 1976;194:1439-41.
Sakakura T, Sakagami Y, Nishizuka Y. Persistence of responsiveness of adult mouse mammary gland to induction by embryonic mesenchyme. Dev Biol 1979;72:201-10.
Satokata I, Ma L, Ohshima H, Bei M, Woo I, Nishizawa K, et al. Msx2 deficiency in mice causes pleiotropic defects in bone growth and ectodermal organ formation. Nat Genet 2000;24:391-5.
Williams JM, Daniel CW. Mammary ductal elongation: differentiation of myoepithelium and basal lamina during branching morphogenesis. Dev Biol 1983;97:274-90.
Gouon-Evans V, Lin EY, Pollard JW. Requirement of macrophages and eosinophils and their cytokines/chemokines for mammary gland development. Breast Cancer Res 2002;4:155-64.
Neville MC, McFadden TB, Forsyth I. Hormonal regulation of mammary differentiation and milk secretion. J Mammary Gland Biol Neoplasia 2002;7:49-66.
Neville MC, Morton J, Umemura S. Lactogenesis. The transition from pregnancy to lactation. Pediatr Clin North Am 2001;48:35-52.
Brisken C, Park S, Vass T, Lydon JP, O'Malley BW, Weinberg RA. Aparacrine role for the epithelial progesterone receptor in mammary gland development. Proc Natl Acad Sci U S A 1998;95:5076-81.
Brisken C, Heineman A, Chavarria T, Elenbaas B, Tan J, Dey SK, et al. Essential function of Wnt-4 in mammary gland development downstream of progesterone signaling. Genes Dev 2000;14:650-4.
Fadok VA. Clearance: the last and often forgotten stage of apoptosis. J Mammary Gland Biol Neoplasia 1999;4:203-11.
Strange R, Metcalfe T, Thackray L, Dang M. Apoptosis in normal and neoplastic mammary gland development. Microsc Res Tech 2001;52:171-81.
Werb Z, Sympson CJ, Alexander CM, Thomasset N, Lund LR, MacAuley A, et al. Extracellular matrix remodeling and the regulation of epithelial-stromal interactions during differentiation and involution. Kidney Int Suppl 1996;54:S68-74.
Michno K, Boras-Granic K, Mill P, Hui CC, Hamel PA. Shh expression is required for embryonic hair follicle but notmammary gland development. Dev Biol 2003;264:153-65.
Gallego MI, Beachy PA, Hennighausen L, Robinson GW. Differential requirements for shh in mammary tissue and hair follicle morphogenesis. Dev Biol 2002;249:131-9.
Andl T, Reddy ST, Gaddapara T, Millar SE. WNT signals are required for the initiation of hair follicle development. Dev Cell 2002;2:643-53.
Kratochwil K, Dull M, Farinas I, Galceran J, Grosschedl R. Lef1 expression is activated by BMP-4 and regulates inductive tissue interactions in tooth and hair development. Genes Dev 1996;10:1382-94.
Mailleux AA, Spencer-Dene B, Dillon C, Ndiaye D, Savona-Baron C, Itoh N, et al. Role of FGF10/FGFR2b signaling during mammary gland development in the mouse embryo. Development 2002;129:53-60.
DasGupta R, Fuchs E. Multiple roles for activated LEF/TCF transcription complexes during hair follicle development and differentiation. Development 1999;126:4557-68.
Petiot A, Conti FJ, Grose R, Revest JM, Hodivala-Dilke KM, Dickson C. A crucial role for Fgfr2-IIIb signalling in epidermal development and hair follicle patterning. Development 2003;130:5493-501.
Stenn KS, Paus R. Controls of hair follicle cycling. Physiol Rev 2001;81:449-94.
Braun KM, Niemann C, Jensen UB, Sundberg JP, Silva-Vargas V, Watt FM. Manipulation of stem cell proliferation and lineage commitment: visualisation of label-retaining cells in wholemounts of mouse epidermis. Development 2003;130:5241-55.
Niemann C, Watt FM. Designer skin: lineage commitment in postnatal epidermis. Trends Cell Biol 2002;12:185-92.
Nanba D, Nakanishi Y, Hieda Y. Role of Sonic hedgehog signaling in epithelial and mesenchymal development of hair follicles in an organ culture of embryonic mouse skin. Dev Growth Differ 2003;45:231-9.
Mill P, Mo R, Fu H, Grachtchouk M, Kim PC, Dlugosz AA, et al. Sonic hedgehog-dependent activation of Gli2 is essential for embryonic hair follicle development. Genes Dev 2003;17:282-94.
Ellis T, Smyth I, Riley E, Bowles J, Adolphe C, Rothnagel JA, et al. Overexpression of Sonic Hedgehog suppresses embryonic hair follicle morphogenesis. Dev Biol 2003;263:203-15.
Chiang C, Swan RZ, Grachtchouk M, Bolinger M, Litingtung Y, Robertson EK, et al. Essential role for Sonic hedgehog during hair follicle morphogenesis. Dev Biol 1999;205:1-9.
Incardona JP, Gaffield W, Kapur RP, Roelink H. The teratogenic Veratrum alkaloid cyclopamine inhibits sonic hedgehog signal transduction. Development 1998;125:3553-62.
Incardona JP, Gaffield W, Lange Y, Cooney A, Pentchev PG, Liu S, et al. Cyclopamine inhibition of Sonic hedgehog signal transduction is not mediated through effects on cholesterol transport. Dev Biol 2000;224:440-52.
Lewis MT, Ross S, Strickland PA, Sugnet CW, Jimenez E, Scott MP, et al. Defects in mouse mammary gland development caused by conditional haploinsufficiency of Patched-1. Development 1999;126:5181-93.
Litingtung Y, Chiang C. Specification of ventral neuron types is mediated by an antagonistic interaction between Shh and Gli3. Nat Neurosci 2000;3:979-85.
Meyer NP, Roelink H. The amino-terminal region of Gli3 antagonizes the Shh response and acts in dorsoventral Hedgehog Network Regulation of Mammary Gland Development 181 fate specification in the developing spinal cord. Dev Biol 2003;257:343-55.
te Welscher P, Zuniga A, Kuijper S, Drenth T, Goedemans HJ, Meijlink F, et al. Progression of vertebrate limb development through SHH-mediated counteraction of GLI3. Science 2002;298:827-30.
Buscher D, Bosse B, Heymer J, Ruther U. Evidence for genetic control of Sonic hedgehog by Gli3 in mouse limb development. Mech Dev 1997;62:175-82.
Brewster R, Mullor JL, Ruiz i Altaba A. Gli2 functions in FGF signaling during antero-posterior patterning. Development 2000;127:4395-405.
Aoto K, Nishimura T, Eto K, Motoyama J. Mouse GLI3 regulates Fgf8 expression and apoptosis in the developing neural tube, face, and limb bud. Dev Biol 2002;251:320-32.
Lewis MT, Ross S, Strickland PA, Sugnet CW, Jimenez E, Hui C, et al. The Gli2 transcription factor is required for normal mouse mammary gland development. Dev Biol 2001;238:133-44.
Goodrich LV, Milenkovic L, Higgins KM, Scott MP. Altered neural cell fates and medulloblastoma in mouse patched mutants. Science 1997;277:1109-13.
Borycki A, Brown AM, Emerson CP, Jr. Shh and Wnt signaling pathways converge to control Gli gene activation in avian somites. Development 2000;127:2075-87.
Silberstein GB, Daniel CW. Investigation of mouse mammary ductal growth regulation using slow-release plastic implants. J Dairy Sci 1987;70:1981-90.
Silberstein GB, Daniel CW. Elvax 40P implants: sustained, local release of bioactive molecules influencingmammaryductal development. Dev Biol 1982;93:272-8.
Silberstein GB, Daniel CW. Reversible inhibition of mammary gland growth by transforming growth factor-beta. Science 1987;237:291-3.
Nilsson M, Unden AB, Krause D, Malmqwist U, Raza K, Zaphiropoulos PG, et al. Induction of basal cell carcinomas and trichoepitheliomas in mice overexpressing GLI-1. Proc Natl Acad Sci U S A 2000;97:3438-43.
Saldanha G. The Hedgehog signalling pathway and cancer. J Pathol 2001;193:427-32.
Watkins DN, Berman DM, Baylin SB. Hedgehog signaling: progenitor phenotype in small-cell lung cancer. Cell Cycle 2003;2:196-8.
Berman DM, Karhadkar SS, Maitra A, Montes De Oca R, Gerstenblith MR, Briggs K, et al. Widespread requirement for Hedgehog ligand stimulation in growth of digestive tract tumours. Nature 2003;425:846-51.
Thayer SP, Di Magliano MP, Heiser PW, Nielsen CM, Roberts DJ, Lauwers GY, et al. Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature 2003;425:851-6.
Barnes EA, Kong M, Ollendorff V, Donoghue DJ. Patched1 interacts with cyclin B1 to regulate cell cycle progression. Embo J 2001;20:2214-23.
Fan H, Khavari PA. Sonic hedgehog opposes epithelial cell cycle arrest. J Cell Biol 1999;147:71-6.
Kenney AM, Rowitch DH. Sonic hedgehog promotes G(1) cyclin expression and sustained cell cycle progression in mammalian neuronal precursors. Mol Cell Biol 2000;20:9055-67.
Long F, Zhang XM, Karp S, Yang Y, McMahon AP. Genetic manipulation of hedgehog signaling in the endochondral skeleton reveals a direct role in the regulation of chondrocyte proliferation. Development 2001;128:5099-108.
Yoon JW, Kita Y, Frank DJ, Majewski RR, Konicek BA, Nobrega MA, et al. Gene expression profiling leads to identification of GLI1-binding elements in target genes and a role for multiple downstream pathways in GLI1-induced cell transformation. J Biol Chem 2002;277:5548-55.
Duman-Scheel M, Weng L, Xin S, Du W. Hedgehog regulates cell growth and proliferation by inducing CyclinDand Cyclin E. Nature 2002;417:299-304.
Aszterbaum M, Rothman A, Johnson RL, Fisher M, Xie J, Bonifas JM, et al. Identification of mutations in the human PATCHED gene in sporadic basal cell carcinomas and in patients with the basal cell nevus syndrome. J Invest Dermatol 1998;110:885-8.
Vorechovsky I, Benediktsson KP, Toftgard R. The patched/hedgehog/smoothened signalling pathway in human breast cancer: no evidence for H133Y SHH, PTCH and SMO mutations. Eur J Cancer 1999;35:711-3.
Wicking C, Evans T, Henk B, Hayward N, Simms LA, Chenevix-Trench G, et al. No evidence for the H133Y mutation in SONIC HEDGEHOG in a collection of common tumour types. Oncogene 1998;16:1091-3.
Hu Z, Bonifas JM, Aragon G, Kopelovich L, Liang Y, Ohta S, et al. Evidence for lack of enhanced hedgehog target gene expression in common extracutaneous tumors. Cancer Res 2003;63:923-8.
Xie J, Johnson RL, Zhang X, Bare JW, Waldman FM, Cogen PH, et al. Mutations of the PATCHED gene in several types of sporadic extracutaneous tumors. Cancer Res 1997;57:2369-72.
Chang-Claude J, Dunning A, Schnitzbauer U, Galmbacher P, Tee L, Wjst M, et al. The patched polymorphism Pro1315Leu (C3944T) may modulate the association between use of oral contraceptives and breast cancer risk. Int J Cancer 2003;103:779-83.
Williams JA, Guicherit OM, Zaharian BI, Xu Y, Chai L, Wichterle H, et al. Identification of a small molecule inhibitor of the hedgehog signaling pathway: Effects on basal cell carcinoma-like lesions. Proc Natl Acad Sci U S A 2003;100:4616-21.
Frank-Kamenetsky M, Zhang XM, Bottega S, Guicherit O, Wichterle H, Dudek H, et al. Small-molecule modulators of Hedgehog signaling: identification and characterization of Smoothened agonists and antagonists. J Biol 2002;1:10.
Chen JK, Taipale J, Young KE, Maiti T, Beachy PA. Small molecule modulation of Smoothened activity. Proc Natl Acad Sci U S A 2002;99:14071-6.
Chen JK, Taipale J, Cooper MK, Beachy PA. Inhibition of Hedgehog signaling by direct binding of cyclopamine to Smoothened. Genes Dev 2002;16:2743-8.
Taipale J, Chen JK, Cooper MK, Wang B, Mann RK, Milenkovic L, et al. Effects of oncogenic mutations in Smoothened and Patched can be reversed by cyclopamine. Nature 2000;406:1005-9.
Berman DM, Karhadkar SS, Hallahan AR, Pritchard JI, Eberhart CG, Watkins DN, et al. Medulloblastoma growth inhibition by hedgehog pathway blockade. Science 2002;297:1559-61.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lewis, M.T., Veltmaat, J.M. Next Stop, the Twilight Zone: Hedgehog Network Regulation of Mammary Gland Development. J Mammary Gland Biol Neoplasia 9, 165–181 (2004). https://doi.org/10.1023/B:JOMG.0000037160.24731.35
Issue Date:
DOI: https://doi.org/10.1023/B:JOMG.0000037160.24731.35