Abstract
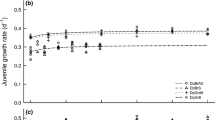
In crustaceans, cholesterol is anEssential nutrient, which they must directly obtain from their food or by bioconversion from other dietary sterols. Eukaryotic phytoplankton contain a great variety of sterols that differ from cholesterol in having additional substituents or different positions and/or number of double bonds in the side chain or in the sterol nucleus. In this study, we investigated to what extent these structural features affect the growth and reproduction of Daphnia galeata in standardized growth experiments with the cyanobacterium Synechococcus elongatus supplemented with single sterols as food source. The results indicated that Δ5 (sitosterol, stigmasterol,Desmosterol) and Δ5,7 (7-dehydrocholesterol, ergosterol) sterols meet the nutritional requirements of the daphnids, while the Δ7 sterol lathosterol supports somatic growth and reproduction to a significantly lower extent than cholesterol. Dihydrocholesterol (Δ0) and lanosterol (Δ8) did not improve the growth of D. galeata, and growth was adversely affected by the Δ4 sterol allocholesterol. Sterols seem to differ in their allocation to somatic growth and reproduction. Thus, structural differences of dietary sterols have pronounced effects on life-history traits of D. galeata.
Similar content being viewed by others
References
Ahlgren, G., Gustafsson, I. B., and Boberg, M. 1992. Fatty acid content and chemical composition of freshwater microalgae. J. Phycol. 28:37–50.
Ahlgren, G., Lundstedt, L., Brett, M. T., and Forsberg, C. 1990. Lipid composition and food quality of some freshwater phytoplankton for cladoceran zooplankters. J. Plankton Res. 12:809–818.
Akihisa, T., Hori, T., Suzuki, H., Sakoh, T., Yokota, T., and Tamura, T. 1992. 24β-Methyl-5α-cholest-9-11-en-3β-ol two 24β-alkyl-δ-5-7-9-11-sterols and other 24β-alkylsterols from Chlorella vulgaris. Phytochemistry 31:1769–1772.
Arts, M. T., Evans, M. S., and Robarts, R. D. 1992. Seasonal patterns of total and energy reserve lipids of dominant zooplanktonic crustaceans from a hyper-eutrophic lake. Oecologia 90:560–571.
Becker, C. and Boersma, M. 2003. Resource quality effects on life histories of Daphnia. Limnol. Oceanogr. 48:700–706.
Behmer, S. T. and Elias, D. O. 2000. Sterol metabolic constraints as a factor contributing to the maintenance of diet mixing in grasshoppers (Orthoptera:Acrididae). Physiol. Biochem. Zool. 73:219–230.
Behmer, S. T. and Grebenok, R. J. 1998. Impact of dietary sterols on life-history traits of a caterpillar. Physiol. Entomol. 23:165–175.
Cobelas, M. A. and Lechardo, J. Z. 1988. Lipids in microalgae. A review I. Biochemistry. Grasas y Aceites 40:118–145.
De Lange, H. J. and Arts, M. T. 1999. Seston composition and the potential for Daphnia growth. Aquatic Ecol. 33:387–398.
DeMott, W. R. and Müller-Navarra, D. C. 1997. The importance of highly unsaturated fatty acids in zooplankton nutrition:evidence from experiments with Daphnia, a cyanobacterium and lipid emulsions. Freshwater Biol. 38:649–664.
Elser, J., Hayakawa, K., and Urabe, J. 2001. Nutrient limitation reduces food quality for zooplankton: Daphnia response to seston phosphorus enrichment. Ecology 82:898–903.
Goad, L. J. 1981. Sterol biosynthesis and metabolism in marine invertebrates. Pure Appl. Chem. 51:837–852.
Grieneisen, M. 1994. Recent advances in our knowledge of ecdysteroid biosynthesis in insects and crustaceans. Insect Biochem. Mol. 24:115–132.
Hai, T., Schneider, B., Schmidt, J., and Adam, G. 1996. Sterols and triterpenoids from the cyanobacterium Anabaena hallensis. Phytochemistry 41:1083–1084.
Harrison, K. E. 1990. The role of nutrition in maturation, reproduction and embryonicDevelopment ofDecapod crustaceans:A review. J. Shellfish Res. 9:1–28.
Harvey, H. R., Eglinton, G., O`hara, S. C. M., and Corner, E. D. S. 1987. Biotransformation and assimilation of dietary lipids by Calanus feeding on a dinoflagellate. Geochim. Cosmochim. Acta 51:3031–3040.
Ikekawa, N. 1985. Structures, biosynthesis and function of sterols in invertebrates, pp. 199–230, in H., Danielsson and J., Sjovall (eds.). Sterols and Bile Acids. Elsevier/North Holland Biomedical, Amsterdam.
Jüttner, F., Leonhardt, J., and Möhren, S. 1983. Environmental factors affecting the formation of mesityloxid, dimethylallylic alcohol and other volatile compounds excreted by Anabaena cylindrica. J. Gen. Microbiol. 129:407–412.
Klein Breteler, W. C. M., Schogt, N., Baas, M., Schouten, S., and Kraay, G. W. 1999. Trophic upgrading of food quality by protozoans enhancing copepod growth: Role ofEssential lipids. Mar. Biol. 135:191–198.
Lachaise, F., Carpentier, G., Somme, G., Colardeau, J., and Beydon, P. 1989. Ecdysteroid synthesis by crab Y-organs. J. Exp. Zool. 252:283–292.
Lampert, W. 1977a. Studies on the carbon balance of Daphnia pulex as related to environmental conditions. I. Methodological problems of the use of 14C for the measurement of carbon assimilation. Arch. Hydrobiol. Suppl. 48:287–309.
Lampert, W. 1977b. Studies on the carbon balance of Daphnia pulex as related to environmental conditions. II. TheDependence of carbon assimilation on animal size, temperature, food concentration and diet species. Arch. Hydrobiol. Suppl. 48:310–335.
Lampert, W. 1981a. Toxicity of the blue-green Microcystis aeruginosa: EffectiveDefense mechanism against grazing pressure by Daphnia. Verh. Int. Verein. Limnol. 21:1436–1440.
Lampert, W. 1981b. Inhibitory and toxic effects of blue-green algae on Daphnia. Rev. Gesamten Hydrobiol. 66:285–298.
Lampert, W. and Trubetskova, I. 1996. Juvenile growth rate as a measure of fitness in Daphnia. Funct. Ecol. 10:631–635.
Müller-Navarra, D. C. 1995. Evidence that a highly unsaturated fatty acid limits Daphnia growth in nature. Arch. Hydrobiol. 132:297–307.
Müller-Navarra, D. C., Brett, M., Liston, A. M., and Goldman, C. R. 2000. A highly unsaturated fatty acid predicts carbon transfer between primary producers and consumers. Nature 403:74–77.
Nes, W. R. and McKean, M. L. 1977. Biochemistry of Steroids and Other Isopentenoids. University Park Press, Baltimore.
Petkov, G. D. and Kim, D. D. 1999. Sterols of the green alga Coelastrum. Algolog. Stud. 130:89–92.
Piironen, V., Lindsay, D., Miettinen, T., Toivo, J., and Lampi, A. M. 2000. Plant sterols: Biosynthesis, biological function and their importance to human nutrition. J. Sci. Food Agric. 80:939–966.
Porter, K. G. and McDonough, R. 1984. The energetic cost of response to blue-green algal filaments by cladocerans. Limnol. Oceanogr. 29:365–369.
Prahl, F. G., Eglinton, G., Corner, E. D. S., O`hara, S. C. M., and Forsberg, T. E. V. 1984. Changes in plant lipids during passage through the gut of Calanus. J. Mar. Biol. Ass. U.K. 64:317–334.
Rees, H. H. 1985. Biosynthesis of ecdysone, pp. 249–293, in G. A., Kerkut and L. I., Gilbert (eds.). Comprehensive Insect Physiology, Biochemistry and Pharmacology. Pergamon, New York.
Rudolph, P. and Spaziani, E. 1992. Formation of ecdysteroids by Y-organs of the crab, Menippe mercenaria: II. Incorporation of cholesterol into 7-dehydrocholesterol and secretion products in vitro. Gen. Comp. Endocr. 88:235–242.
Rudolph, P., Spaziani, E., and Wang, W. 1992. Formation of ecdysteroids by Y-organs of the crab, Menippe mercenaria: I. Biosynthesis of 7-dehydrocholesterol in vivo. Gen. Comp. Endocr. 88:224–234.
Stich, H.-B. and Lampert, W. 1984. Growth and reproduction of migrating and non-migrating Daphnia species under simulated food and temperature conditions of diurnal vertical migration. Oecologia 61:192–196.
Svoboda, J. A. and Thompson, M. J. 1985. Steroids, pp. 137–175, in G. A., Kerkut and L. I., Gilbert (eds.). Comprehensive Insect Physiology, Biochemistry and Pharmacology. Pergamon, New York.
Teshima, S. 1971. Bioconversion of β-sitosterol and 24-methylcholesterol to cholesterol in marine crustacea. Comp. Biochem. Physiol. B 39:815–822.
Teshima, S. and Kanazawa, A. 1971a. Sterol compositions of marine crustaceans. Bull. Jpn. Soc. Sci. Fish. 37:63–67.
Teshima, S. and Kanazawa, A. 1971b. Bioconversion of the dietary ergosterol to cholesterol in Artemia salina. Comp. Biochem. Physiol. B 38:603–607.
Teshima, S. and Kanazawa, A. 1973. Metabolism ofDesmosterol in the prawn, Penaeus japonicus. Mem. Fac. Fish. Kagoshima Univ. 22:15–19.
Teshima, S., Kanazawa, A., and Sasada, H. 1983. Nutritional value of dietary cholesterol and other sterols to larval prawn, Penaeus japonicus Bate. Aquaculture 31:159–167.
Urabe, J. and Sterner, R. W. 2001. Contrasting effects of different types of resourceDepletion on life-history traits in Daphnia. Funct. Ecol. 15:165–174.
Volkman, J. K. 2003. Sterols in microorganisms. Appl. Microbiol. Biotech. 60:495–506.
Von Elert, E. 2002. Determination of limiting polyunsaturated fatty acids in Daphnia galeata using a new method to enrich food algae with single fatty acids. Limnol. Oceanogr. 47:1764–1773.
Von Elert, E., Martin-Creuzburg, D., and Le Coz, J. R. 2003. Absence of sterols constrains carbon transfer between cyanobacteria and a freshwater herbivore (Daphnia galeata. Proc. R. Soc. Lond. B Biol. 270:1209–1214.
Von Elert, E. and Wolffrom, T. 2001. Supplementation of cyanobacterial food with polyunsaturated fatty acids does not improve growth of Daphnia. Limnol. Oceanogr. 46:1552–1558.
Wacker, A. and Von Elert, E. 2001. Polyunsaturated fatty acids: Evidence for non-substitutable biochemical resources in Daphnia galeata. Ecology 82:2507–2520.
Yasuda, S. 1973. Sterol compositions of crustaceans. I. Marine and fresh-waterDecapods. Comp. Biochem. Physiol. B 44:41–46.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Martin-Creuzburg, D., Elert, E.V. Impact of 10 Dietary Sterols on Growth and Reproduction of Daphnia galeata . J Chem Ecol 30, 483–500 (2004). https://doi.org/10.1023/B:JOEC.0000018624.94689.95
Issue Date:
DOI: https://doi.org/10.1023/B:JOEC.0000018624.94689.95