Abstract
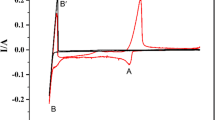
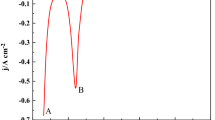
The electrode reactions of the Np3+/Np couple at liquid Cd and Bi electrodes were investigated by cyclic voltammetry at 723, 773 and 823 K in the LiCl–KCl eutectic melt. It was found that the diffusion of Np3+ in the salt phase was the rate-determining step in the cathodic reaction when the concentration of NpCl3 was less than about 1 wt % and the liquid Cd or Bi phase was not saturated with Np. The redox potentials of the Np3+/Np couple at the liquid Cd electrode at 723, 773 and 823 K were observed to be more positive than those at the Mo electrode by 0.158, 0.140 and 0.126 V, respectively. The potential shift results from a lowering of the activity of Np in the Cd phase according to the formation of the NpCd11 alloy at 723 K and NpCd6 at 773 and 823 K. The redox potentials of the Np3+/Np couple at the liquid Bi electrode at 723, 773 and 823 K were more positive than those at the Mo electrode by 0.427, 0.419 and 0.410 V, respectively, which is attributable to a lowering of the activity of Np in the Bi phase due to the formation of NpBi2.
Similar content being viewed by others
References
Y.I. Chang, Nucl. Technol. 88 (1989) 129.
J.P. Ackerman, Ind. Eng. Chem. Res. 30 (1991) 141.
M. Iizuka, T. Koyama, N. Kondo, R. Fujita and H. Tanaka, J. Nucl. Mater. 247 (1997) 183.
Y. Arai, T. Iwai, K. Nakajima and Y. Suzuki, Proceedings of the International Conference on 'Future Nuclear Systems' (GLOBAL' 97), Vol. 1, Yokohama, Japan, 5-10, Oct. 1997, (1997), p. 664.
T. Ogawa, M. Akabori, Y. Suzuki, F. Kobayashi, T. Osugi and T. Mukaiyama, Proceedings, vol. 1, ref. [4], p. 812.
P. Chiotti, V.V. Akhachinskij, I. Ansara and M.H. Rand, in V. Medvedev et al. (Eds), The Chemical Thermodynamics of Actinide Elements and Compounds, Part 5 The Actinide Binary Alloys (International Atomic Energy Agency, Vienna, 1981).
J.P. Ackerman and J.L. Settle, J. Alloys Comp., 199 (1993) 77.
T. Koyama, T.R. Johnson and D.F. Fischer, J. Alloys Comp. 189 (1992) 37.
M. Kurata, Y. Sakamura, T. Hijikata and K. Kinoshita, J. Nucl. Mater. 227 (1995) 110.
H. Moriyama, H. Yamana, S. Nishikawa, S. Shibata, N. Wakayama, Y. Miyashita, K. Moritani and T. Mitsugashira, J. Alloys Comp. 271-273 (1998) 587.
O. Shirai, M. Iizuka, T. Iwai, Y. Suzuki and Y. Arai, J. Electroanal. Chem. 490 (2000) 31.
O. Shirai, M. Iizuka, T. Iwai, Y. Suzuki and Y. Arai, Anal. Sci. 17 (2001) 51.
O. Shirai, K. Uozumi, T. Iwai and Y. Arai, Anal. Sci. 17 (2002) 1959.
M. Krumpelt, I. Johnson and J.J. Heiberger, J. Less-Common Metals 18 (1969) 35.
M. Krumpelt, I. Johnson and J.J. Heiberger, Metall. Trans. 5 (1974) 65.
Y. Sakamura, O. Shirai, T. Iwai and Y. Arai, J. Electrochem. Soc. 147 (2000) 642.
O. Shirai, M. Iizuka, T. Iwai and Y. Arai, J. Appl. Electrochem. 31 (2001) 1055.
M.A. Lewis and T.R. Johnson, J. Electrochem. Soc. 137 (1990) 1414.
A.J. Bard and L.R. Faulkner, 'Electrochemical Methods: Fundamentals and Applications' (John Wiley & Sons, New York 1980), chapter 6.
D.K. Gosser, Jr, 'Cyclic Voltammetry: Simulation and Analysis of Reaction Mechanisms' (VCH, New York, 1993), chapter 2.
I. Johnson, M.G. Chasanov and R.M. Yonco, Trans. Met. Soc. AIME 233 (1965) 1408.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shirai, O., Uozumi, K., Iwai, T. et al. Electrode reaction of the Np3+/Np couple at liquid Cd and Bi electrodes in LiCl–KCl eutectic melts. Journal of Applied Electrochemistry 34, 323–330 (2004). https://doi.org/10.1023/B:JACH.0000015615.17281.51
Issue Date:
DOI: https://doi.org/10.1023/B:JACH.0000015615.17281.51