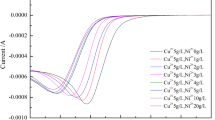
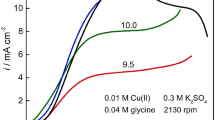
Abstract
When using a copper working electrode to study the diffusion of Cu(II) ions it is necessary to polish the electrode between experiments to maintain a smooth surface. To eliminate the required polishing, a platinum working electrode can be used. The copper layer established on the electrode surface during experimentation can be removed between experiments by anodic stripping. When using a platinum disk working electrode, the most reliable method of analysis involves double potential step chrono-amperometry method as opposed to voltammetry. By applying this method diffusion coefficients for Cu(II) ions in sulphuric acid – aqueous and methanesulfonic acid (HMSA) – methanol solutions were determined.
Similar content being viewed by others
References
T.I. Quickenden and Q. Xu, J. Electrochem. Soc. 143 (1996) 1248.
T.I. Quickenden and X. Jiang, Electrochim. Acta 29 (1984) 693.
L. Grambow and W. Vielstich, J. Electrochem. Soc. 12 (1974) 779.
L.J.J. Janssen, J. Appl. Electrochem. 18 (1988) 339.
M-C. Petit, Electrochim. Acta 10 (1964) 291.
A.J. Bard and L.R. Faulkner, 'Electrochemical Methods' (John Wiley and Sons, New York, 1980).
R.N. Adams, 'Electrochemistry at Solid Electrodes' (Marcel Dekker, New York, 1969), p. 92.
C.J. Milora, J.F. Henrickson and W.C. Hahn, J. Electrochem. Soc. 120 (1973) 488.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
MacHardy, S., Janssen, L. The diffusion coefficient of Cu(II) ions in sulfuric acid–aqueous and methanesulfonic acid–methanol solutions. Journal of Applied Electrochemistry 34, 169–174 (2004). https://doi.org/10.1023/B:JACH.0000009956.75577.ef
Issue Date:
DOI: https://doi.org/10.1023/B:JACH.0000009956.75577.ef