Abstract
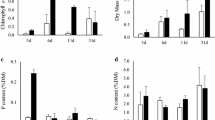
Colonisation of epiphytic algae on the common reed (Phragmites australis) and on glass slides were studied during a twenty-four week exposure period in a shallow, turbid lake, Manyas (Bird Paradise), western Turkey. Slides were used as substrate and positioned near the reed stands. To understand the effect of light on the colonisation of periphyton on P. australis and artificial substrates, three perspex frames (each frame consisted of 60 glass slides) were positioned at depths of ca. 25 cm, 50 cm and 75 cm, called `upper', `middle', and `lower section', below the water surface. For the determination of periphyton and chlorophyll a content three new stems, which were cut above the rhizomes and subdivided into 25 cm, and three slides were taken from the each frame at four-weeks intervals. The rapid increase was recorded in algal species richness and biovolume on P. australis, whereas relatively slow colonisation was observed on glass slides. Filamentous centric diatoms, (mainly Aulacoseira italica and Melosira varians) was the dominant group, accounting for 29 to 82% of the total algal biovolume, and adnate diatoms (Surirella spp.), filamentous, and Chlorococcal chlorophytes (Pediastrum spp. and Oedogonium spp.) were the subdominant groups on the artificial substrate during the colonisation period. Pennate diatoms, mainly prostrate/motile and filamentous diatoms (Navicula tripunctata, Navicula radiosa, and Aulacoseira muzzanensis), and filamentous cyanobacterium (Planktolyngbya limnetica) were recorded as dominant groups on the reeds. Generally, epiphytic algal biomass was higher upon P. australis than upon glass slides. However, after the twelfth week colonisation biomass increased less than on earlier stages on the P. australis. Total algal biovolume was positively correlated with chlorophyll a and with Soluble Reactive Phosphorus (SRP), and negatively correlated with the water level and Secchi disc depth.
Similar content being viewed by others
References
Ács, É., A. K. Borsodi, J. Makk, P. Molnár, A. Mózes, A. Rusznydk, M. N. Reskóné & K. T. Kiss, 2003. Algological and bacteriological investigations on reed periphyton in Lake Velencei, Hungary. Hydrobiologia 506–509: 549–557.
Ács, É. & K. Buczkó, 1994. Daily changes of reed periphyton composition in a shallow Hungarian lake (Lake Velencei). 13th International Diatom Symposium: 1–10.
Anagnostidis, K. & J. Komárek, 1988. Modern approach to the classification system of Cyanophytes, 3. Oscillatoriales. Arch. Hydrobiol. 80: 327–472.
APHA – AWWA –WPCF, 1989. Standard Methods for the Examination ofWater andWastewater, 17th ed., American Public Health Association, Washington, D.C.
Balik, S. & M. R. Ustaoglu, 1990. Investigation of the bioecological features of common carp (Cyprinus carpio L., 1758) population in Lake Manyas, Bandirma. X. National Biology Congress, 18–20 July, Erzurum, Turkey: 271–282 (in Turkish).
Biggs, B. J. F. 1995. The contribution of flood disturbance, catchment geology and land use to the habitat template of periphyton in stream ecosystem. Freshwat. Biol. 33: 419–438.
Boston, H. L. & W. R. Hill, 1991. Photosynthesis–light relations of stream periphyton communities. Limnol. Oceanogr. 36: 644– 656.
Buczkó, K. & É. Ács, 1996. Zonation of periphytic algae in two Hungarian shallow lakes (Lake Velencei and Fertö). Acta Bot. Hung. 40: 21–34.
Buczkó, K. & É. Ács, 1998. Comparison of succession of reed periphyton in a degraded and in an undisturbed part of a shallow lake (Lake Velencei, Hungary, Central Europe). Verh. int. Ver. theor. angew. Limnol. 26: 1674–1676.
Buyukisik, B. & H. Parlak, 1989. Investigation on ecology of the Bird lake Bandirma, A. U. J. Fisher. 6(21-22-23-24): 160–175 (in Turkish).
Cattaneo, A., G. Galanti., S. Gentinetta & S. Romo, 1998. Epiphytic algae and macroinvertebrates on submerged and floating-leaved macrophytes in an Italian lake. Freshwat. Biol. 39: 725–740.
Cottingham, K. L. & S. R. Carpenter, 1998. Population, community, and ecosystem variates as ecological indicators; phytoplankton responses to whole-lake enrichment. Ecol. Appl. 8: 508–530.
Cronk, J. K. & W. J. Mitsch, 1994. Periphyton on artificial and natural surfaces in constructed freshwater wetlands under different hydrologic regimes. Aquat. Bot. 48: 325–341.
Goldsborough, L. G. & M. Hickman, 1991. A comparison of periphytic algal biomass and community structure on Scirpus validus and on a morphologically similar artificial substratum. J. Phycol. 27: 916–207.
Hillebrand, H. & U. Sommer, 2000. Diversity of benthic microalgae in response to colonisation time and eutrophication. Aquat. Bot. 67: 221–236.
Howard-Williams, C. & B. R. Davies, 1978. The influence of periphyton on the surface structure of a Potamogeton pectinatus L. leaf (a hypothesis). Aquat. Bot. 5: 87–91.
Huber-Pestalozzi, G., 1969. Das phytoplankton des süsswassers. Euglenophyceen. 4. Teil, Band XVI.
Hustedt, F., 1985. The Pennate Diatoms. Koeltz Scientific Books Koenigstein. Supplement by G. Jensen.
Komárek, J. & B. Fott, 1983. Chlorophyceae, (Grünalgen), Ordnung: Chlorococcales. In Das Phytoplankton des Süsswassers (founded by G. Huber-Pestalozzi). Die Binnengewasser 16, 7/1. E. Schweizerbart'sch Verlagsbuchhandlung, Stuttgart: 1044 pp.
Komárek, J. & K. Anagnostidis, 1986. Modern approach to the classification system of Cyanophytes. 2. Chroococcales. Arch. Hydrobiol. Suppl. 73: 157–226.
Koray, T. 1987. Comparison of the diversity index to determine the changes of the phytoplankton assemblages. Do?a TU Müh. ve Çev. D.C. 11(2): 242–253 (in Turkish).
Lowe, R. L., J. B. Guckert, S. E. Belanger, D. H. Davidson & D.W. Johnson, 1996. An evaluation of periphyton community structure and function on tile and cobble substrate in experimental stream mesocosms. Hydrobiologica 328: 135–146.
Meulemans, J. T., 1988. Seasonal changes in biomass and production of periphyton growing upon reed in Lake Maarseven I. Arch. Hydrobiol. 112: 21–42.
Müller, U., 1995. Vertical zonation and production rates of epiphytic algae on Phragmites australis. Freshwat. Biol. 34: 69–80.
Müller, U., 1996. Production rates of epiphytic algae in a eutrophic lake. Hydrobiologia 330: 37–45.
Müller, U., 1999. The vertical zonation of adpressed diatoms and other epiphytic algae on Phragmites australis. Eur. J. Phycol. 34: 487–496.
Nusch, E., 1980. Comparison of different methods for chlorophyll and phaeopigment determination. Arch. Hydrobiol. Beih. Ergebn. Limnol. 14: 14–36.
Patrick, R. & C.W. Reimer, 1966. The Diatoms of the United States, Vol. 1. The Academy of Natural Sciences of Philadephia.
Patrick, R. & C.W. Reimer, 1975. The Diatoms of the United States, Vol. 2, Part 1. The Academy of Natural Sciences of Philadephia.
Romo, S. & G. Galanti, 1998. Vertical and seasonal distribution of epiphytic algae on water chestnut (Trapa natans). Arch. Hydrobiol. 141: 483–504.
Rott, E., 1981. Some results from phytoplankton counting intercalibrations. Schweizerische Zeitschrift für Hydrologie – Swiss J. Hydrol. 43: 34–62.
Szabó, K., É. Ács, E. Pápista, K. T. Kiss, S. Barreto & J. Makk, 2001. Periphyton and phytoplankton in the Soroksár – Danube in Hungary. I. Periphytic algae on reed stems. Acta Bot. Hung. 43: 13–35.
Utermöhl, H., 1958. Zur Vervollkommung der quantitativen phytoplankton-methodik. Mitteil. int. Ver. theor. angew. Limnol. 9: 39 pp.
Van Dijk, G. M., 1993. Dynamics and attenuation characteristic of periphyton upon artifical substratum under various light conditions and some additional observations on periphyton upon Potamogeton pectinatus L. Hydrobiologia 252: 143–161.
Vinebrooke, R. D. & P. R. Leavitt, 1996. Effects of ultraviolet radiation on periphyton in an alpine lake. Limnol. Oceanogr. 41: 1035–1040.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Albay, M., Akcaalan, R. Comparative study of periphyton colonisation on common reed (Phragmites australis) and artificial substrate in a shallow lake, Manyas, Turkey. Hydrobiologia 506, 531–540 (2003). https://doi.org/10.1023/B:HYDR.0000008606.69572.f6
Issue Date:
DOI: https://doi.org/10.1023/B:HYDR.0000008606.69572.f6