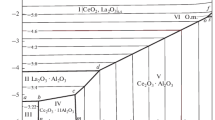
Abstract
The kinetic specifics of the oxidation of alloyed steel under a protective layer containing iron oxide are considered. The dependence of the steel oxidation rate on the concentration of Fe2O3 in the melt is experimentally determined.
Similar content being viewed by others
REFERENCES
P. I. Buler, O. R. Ivanova, and G. R. Zarina, “The effect of moisture on the rate of iron oxidation under the effect of a glass-enamel melt,” Zashch Met., 19(4), 654–658 (1983).
P. I. Buler and O. R. Lazutkina, “Propagation of corrosion in steel under a melted glass film in air,” Zashch. Met., 20(3), 439–442 (1984).
P. I. Buler, O. R. Lazutkina, E. A. Zubkova, and S. P. Panov, “Oxidation of steel 30KhGSA in borosilicate melt containing Fe2 O3,” in: Physiochemical Studies of Metallurgical Processes, Issue 14[in Russian] (1989), pp. 102–108.
P. I. Buler, O. R. Lazutkina, Z. A. Tagintseva, and V. A. Yushkova, “Oxidation of iron and cobalt under a layer of protective film of aluminoborosilicate melt,” in: Proc. 5th All-Union Conf. on Structure and Properties of Metallic and Slag Melts[in Russian], Sverdlovsk, (1983), pp. 94–96.
O. R. Lazutkina, “Development and study of a glass lubricant based on industrial waste,” Stroitel'stvo Obrazovanie, Issue 6, 217–219 (2003).
É. A. Pastukhov, O. A. Esin, and N. A. Vatolin, Physicochemical Principles of Metallurgical Processes[in Russian], Nauka, Moscow (1969).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lazutkina, O.R., Buler, P.I. Oxidation of Steel 30KhGSA in Borosilicate Melt Containing Iron Oxides. Glass and Ceramics 61, 206–207 (2004). https://doi.org/10.1023/B:GLAC.0000043095.01811.86
Issue Date:
DOI: https://doi.org/10.1023/B:GLAC.0000043095.01811.86