Abstract
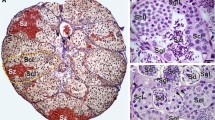
The morphometric study of spermatogenic cysts in sexually mature tilapias, during the evolution of spermatogenesis, showed a dramatic increase in both number of germ cells and cyst volume. However, the opposite trend was observed for germ cell size. Nevertheless, the number of Sertoli cells increased gradually up to leptotene/zygotene cysts, stabilizing thereafter. Based on the number of spermatids supported by each Sertoli cell and compared to mammals, Sertoli cell efficiency in tilapias is remarkably high. Sertoli cell proliferation was frequently observed, mainly in spermatogonial cysts, and probably is the major factor related to the testis growth and the increase in sperm production that normally occurs in adult tilapias. The combined duration of spermatocytes (5 days) and spermiogenic (5–6 days) phases of spermatogenesis in fish kept at 25 °C was 10–11 days. Mainly due to acceleration in meiosis, these two phases lasted a total of 6 days in tilapias kept at 30 °C, in the opposite way, at 20 °C spermatogenesis was arrested at pachytene spermatocytes. To our knowledge, this is the most comprehensive investigation performed up to date on testis morphometry and function in adult tilapias.
Similar content being viewed by others
References
Billard, R. 1969. La spermatogenèse de Poecilia reticilata. IEstimation du nombre de génerations goniales et rendement de la spermatogenése. Ann. Biol. Bioch. Biophys. 9: 251–271.
Billard, R. 1990. Spermatogenesis in teleost fish. In: Reproduction in the male. pp. 183–212. Edited by G.E. Lamming, Churchill Livingstone.
Cobb, J., Cargile, B. and Handel, M.A. 1999. Acquisition of competence to condense metaphase I chromosomes during spermatogenesis. Dev. Biol. 205: 49–64.
Eddy, E.M. 2002. Male germ cell gene expression. Recent Prog. Horm. Res. 57: 103–28.
Egami, N. and Hyodo-Taguchi, Y 1967. An autoradiographic examination of rate of spermatogenesis at different temperatures in the fish, Oryzias latipes. Exp. Cell Res. 47: 665–667.
França, L.R. and Russell, L.D. 1998. The testis of domestic animals. In: Male reproduction: a multidisciplary overview. pp. 198–219. Edited by F. Martínez-García and J. Regadera, Churchill Communications, Madrid.
França, L.R., Ogawa, T., Avarbock, M.R., Brinster, R.L. and Russell, L.D. 1998. Germ cell genotype controls cell cycle during spermatogenesis in the rat. Biol. Reprod. 59: 1371–1377.
França, L.R., Silva Jr., V.A., Chiarini-Garcia, H., Garcia, S.K. and Debeljuk, L. 2000. Cell proliferation and hormonal changes during postnatal development of the testis in the pig. Biol. Reprod. 63: 629–1636.
Koulish, S., Kramer, C.R. and Grier, H.J. 2002. Organization of the male gonad in a protogynous fish, Thalassoma bifasciatum (Teleostei: Labridae). J. Morphol. 254: 292–311.
Liu, D., Liao, C. and Wolgemuth, D.J. 2000. A role for cyclin A1 in the activation of MPF and G2-M transition during meiosis of male germ cells in mice. Dev. Biol. 224: 388–400.
Shimizu, A. 2003. Effect of photoperiod and temperature on gonadal activity and plasma steroid levels in a reared strain of the mummichog (Fundulus heteroclitus) during different phases of its annual reproductive cycle. Gen. Comp. Endocr. 131: 310–324.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vilela, D., Silva, S., Peixoto, M. et al. Spermatogenesis in teleost: insights from the Nile tilapia (Oreochromis niloticus) model. Fish Physiology and Biochemistry 28, 187–190 (2003). https://doi.org/10.1023/B:FISH.0000030523.16010.62
Issue Date:
DOI: https://doi.org/10.1023/B:FISH.0000030523.16010.62