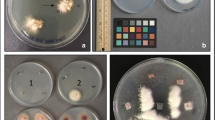
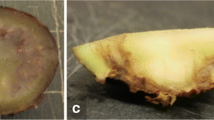
Abstract
The main causative agents of Fusarium head blight are Fusarium graminearum and Fusarium culmorum. We examined the mycotoxin-producing abilities and molecular variability of 37 Fusarium culmorum isolates collected from the Pan-Northern Hemisphere, together with isolates representing related species. Mycotoxin-producing abilities of the isolates were tested by thin layer chromatography and by PCR using primer pairs specific for the Tri7 and Tri13 genes. Thirty isolates belonged to chemotype I (producing deoxynivalenol and 3-acetyl-deoxynivalenol), while seven represented chemotype II (producing nivalenol and/or fusarenone X). The presence of a functional Tri7 gene correlated well with nivalenol production. Isolates belonging to chemotype I were in general more pathogenic in in vitro tests than those belonging to chemotype II. Phylogenetic analysis of the random amplified polymorphic DNA profiles (RAPD) of the isolates enabled the isolates to be clustered into different groups. Most isolates from Hungary exhibited identical RAPD profiles. A similar clustering was found on the tree based on restriction analysis of the intergenic spacer region data. Sequence analysis of a putative reductase gene fragment of the isolates was also carried out. A correlation was detected between the geographic origin of the isolates and their position on the cladogram produced based on sequence data. The presence of mating type gene homologues was also tested with primer pairs specific for MAT1-1 and MAT1-2. The isolates carried either MAT1-1 or MAT1-2 homologues. No correlation was observed between clustering of the isolates based on RAPD, restriction analysis of the intergenic spacer region or sequence data and the distribution of MAT idiomorphs. Similarly, no correlation was detected between mycotoxin-producing abilities or aggressiveness and molecular characteristics of the isolates. Statistical analysis of RAPD data and lack of strict correlation between trees based on different data sets supported the view that Fusarium culmorum has a recombining population structure. The presence of mating type gene homologues in the isolates indicates that the recombining population structure is caused by ongoing or past meiotic exchanges.
Similar content being viewed by others
References
Abramson D, Clear RM, Gaba D, Smith DM, Patrick SK and Saydak D (2001) Trichothecene and moniliformin production by Fusarium species from western Canadian wheat. Journal of Food Protection 64: 1220-1225
Agapow PM and Burt A (2001) Indices of multilocus linkage disequilibrium. Molecular Ecology Notes 1: 101-102
Alexander NJ, McCormick SP and Ziegenhorn SL (2000) Phytotoxicity of selected trichothecenes using Chlamydomonas reinhardtii as a model system. Natural Toxins 7: 265-269
Appel DJ and Gordon TR (1995) Intraspecific variation within populations of Fusarium oxysporum based on RFLP analysis of the intergenic spacer region of the rDNA. Experimental Mycology 19: 120-128
Bai GH, Desjardins AE and Plattner RD (2001) Deoxynivale-nol non-producing Fusarium graminearum causes initial infection, but does not cause disease spread in wheat spikes. Mycopathologia 153: 91-98
Bakan B, Giraud-Delville C, Pinson L, Richard MD, Fournier E and Brygoo Y (2002) Identification by PCR of Fusarium culmorum strains producing large and small amounts of deoxynivalenol. Applied and Environmental Microbiology 68: 5472-5479
Bakan B, Pinson L, Cahagnier B, Melcion D, Semon E and Richard MD (2001) Toxigenic potential of Fusarium culmorum strains isolated from French wheat. Food Additives and Contaminants 18: 998-1003
Birzele B, Meier A, Hindorf H, Kramer J and Dehne HW (2002) Epidemiology of Fusarium infection and deoxynivalenol content in winter wheat in the Rhineland, Germany. European Journal of Plant Pathology 108: 667-673
Bottalico A and Perrone G (2002) Toxigenic Fusarium species and mycotoxins associated with head blight in small-grain cereals in Europe. European Journal of Plant Pathology 108: 611-624
Brown DW, McCormick SP, Alexander NJ, Proctor RH and Desjardins AE (2002) Inactivation of a cytochrome P-450 is a determinant of trichothecene diversity in Fusarium species. Fungal Genetics and Biology 36: 224-233
Buntjer JB (1997) Phylogenetic computer tools (PhylTools), Version 1.32 for Windows. Laboratory of Plant Breeding, Wageningen University, The Netherlands
Burt A, Carter DA, Koenig GL, White TJ and Taylor JW (1996) Molecular markers reveal cryptic sex in the human pathogen Coccidioides immitis. Proceedings of the National Academy of Sciences USA 93: 770-773
Carter JP, Rezanoor HN, Holden D, Desjardins AE, Plattner RD and Nicholson P (2002) Variation in pathogenicity associated with the genetic diversity of Fusarium graminearum. European Journal of Plant Pathology 108: 573-583
De Nijs M, Larsen JS, Gams W, Rombouts FM, Wernars K, Thrane U and Notermans SHW (1997) Variations in random amplified polymorphic DNA patterns and secondary metabolite profiles within Fusarium species from cereals from various parts of The Netherlands. Food Microbiology 14: 449-457
Dover GA (1982) Molecular drive: A cohesive mode of species evolution. Nature 299: 111-117
Edel V, Steinberg C, Gautheron N, Recorbet G and Alabouvette C (2001) Genetic diversity of Fusarium oxysporum populations isolated from different soils in France. FEMS Microbiology Ecology 36: 61-71
Eudes F, Comeau A, Rioux S and Collin J (2001) Impact of trichothecenes on Fusarium head blight (Fusarium graminearum) development in spring wheat (Triticum aestivum). Canadian Journal of Plant Pathology 23: 318-322
Eudes FJS, Collin J, Rioux S and Comeau A (1997) The trichothecenes, a major component of wheat pathogenesis. Cereal Research Communications 25: 495-496
Felsenstein J (1985) Confidence limits on phylogenies: An approach using the bootstrap. Evolution 39: 783-791
Felsenstein J (1995) PHYLIP (Phylogeny Inference Package) Version 3.57c. Department of Genetics, University of Washington, Seattle, USA
Gang G, Miedaner T, Schuhmacher U, Schollenberger M and Geiger HH (1998) Deoxynivalenol and nivalenol production by Fusarium culmorum isolates differing in aggressiveness toward winter rye. Phytopathology 88: 879-884
Hestbjerg H, Felding G and Elmholt S (2002a) Fusarium culmorum infection of barley seedlings: Correlation between aggressiveness and deoxynivalenol content. Journal of Phytopathology 150: 308-312
Hestbjerg H, Nielsen KF, Thrane U and Elmholt S (2002b) Production of trichothecenes and other secondary metabolites by Fusarium culmorum and Fusarium equiseti on common laboratory media and a soil organic matter agar: An ecological interpretation. Journal of Agricultural and Food Chemistry 50: 7593-7599
James TY, Moncalvo JM, Li S and Vilgalys R (2001) Polymorphism at the ribosomal DNA spacers and its relation to breeding structure of the widespread mushroom Schizophyllum commune. Genetics 157: 149-161
Kerényi Z and Hornok L (2002) Structure and function of mating-type genes in Fusarium species. Acta Microbiologica et Immunologica Hungarica 49: 313-314
Kerényi Z, Mule G, Waalwijk C, Oláh B and Hornok L (in press) Mating type sequences in asexually reproducing Fusarium species. Applied and Environmental Microbiology
Kim JC, Kang HJ, Lee DH, Lee YW and Yoshizawa T (1993) Natural occurrence of Fusarium mycotoxins (trichothecenes and zearalenone) in barley and corn in Korea. Applied and Environmental Microbiology 59: 3798-3802
Kimura M (1980) A simple method for estimating evolutionary rate of base substitutions through comparative studies on nucleotide sequences. Journal of Molecular Evolution 2: 87-90
Láday M and Szécsi A (2001) Distinct electrophoretic isozyme profiles of Fusarium graminearum and closely related species. Systematic and Applied Microbiology 24: 67-75
Langseth W, Ghebremeskel M, Kosiak B, Kolsaker P and Miller D (2000) Production of culmorin compounds and other secondary metabolites by Fusarium culmorum and F. graminearum strains from Norwegian cereals. Mycopathologia 152: 23-34
Lauren DR, Sayer ST and Di Menna ME (1992) Trichothecene production by Fusarium species isolated from grain and pasture throughout New Zealand. Mycopathologia 120: 167-176
Leach J, Finkelstein DB and Rambosek JA (1986) Rapid miniprep of DNA from filamentous fungi. Fungal Genetics Newsletter 33: 32-33
Lee T, Han YK, Kim KH, Yun SH and Lee YW (2002) Tri13 and Tri7 determine deoxynivalenol and nivalenol-producing chemotypes of Gibberella zeae. Applied and Environmental Microbiology 68: 2148-2154
Mesterházy Á(1985) Effect of seed production area on the seedling resistance of wheat to Fusarium seedling blight. Agronomie 5: 491-497
Mesterházy Á (2002) Role of deoxynivalenol in aggressiveness of Fusarium graminearum and F. culmorum and in resistance to Fusarium head blight. European Journal of Plant Pathology 108: 675-684
Miedaner T and Reinbrecht C (2001) Trichothecene content of rye and wheat genotypes inoculated with a deoxynivalenol-and a nivalenol-producing isolate of Fusarium culmorum. Journal of Phytopathology 149: 245-251
Miedaner T, Schilling AG and Geiger HH (2001) Molecular genetic diversity and variation for aggressiveness in populations of Fusarium graminearum and Fusarium culmorum sampled from wheat fields in different countries. Journal of Phytopathology 149: 641-648
Miller JD, Greenhalgh R, Wang YZ and Lu M (1991) Trichothecene chemotypes of three Fusarium species. Mycologia 83: 121-130
Miller RNG, Soarea AMQ and Lopes CA (1999) Molecular comparison of Fusarium populations causing eumartii wilt and dry rot of potato in Brazil. Fitopatologica Brasiliensis 24: 149-155
Mills JT (1989) Ecology of mycotoxigenic Fusarium species on cereal seeds. Journal of Food Protection 52: 737-742
Mirocha CJ, Xie W, Xu Y, Wilcoxson RD, Woodward RP, Etebarian RH and Behele G (1994) Production of trichothecene mycotoxins by Fusarium graminearum and F. culmorum on barley and wheat. Mycopathologia 128: 19-23
Mishra PK, Fox RTV and Culham A (2000) Application of nr-DNA ITS sequence for identification of Fusarium culmorum isolates. EPPO Bulletin 30: 493-498
Mishra PK, Fox RTV and Culham A (2002) Restriction analysis of PCR amplified nrDNA regions revealed intraspecific variation within populations of Fusarium culmorum. FEMS Microbiology Letters 215: 291-296
Muthomi JW, Schutze A, Dehne HW, Mutitu EW and Oerke EC (2000) Characterization of Fusarium culmorum isolates by mycotoxin production and aggressiveness to winter wheat. Journal of Plant Disease and Protection 107: 113-123
O'Donnell K, Kistler HC, Tacke BK and Casper HH (2000) Gene genealogies reveal global phylogeographic structure and reproductive isolation among lineages of Fusarium graminearum, the fungus causing wheat scab. Proceedings of the National Academy of Sciences USA 97: 7905-7910
Proctor RH, Desjardins AE, McCormick SP, Plattner RD, Alexander NJ and Brown DW (2002) Genetic analysis of the role of trichothecene and fumonisin mycotoxins in the virulence of Fusarium. European Journal of Plant Pathology 108: 691-698
Rinyu E, Varga J and Ferenczy L (1995) Phenotypic and genotypic analysis of variability in Aspergillus fumigatus. Journal of Clinical Microbiology 33: 2567-2575
Rotter BA, Prelusky DB and Pestka JJ (1996) Toxicology of deoxynivalenol (vomitoxin). Journal of Toxicology and Environmental Health 48: 1-34
Ryu JC, Ohtsubo K, Izumiyama N, Nakamura K, Tanaka T, Yamamura H and Ueno Y (1988) The acute and chronic toxicities of nivalenol in mice. Fundamental and Applied Toxicology 11: 38-47
Saitou N and Nei M (1987) The neighbor-joining method: A new method for reconstructing phylogenetic trees. Molecular Biology and Evolution 4: 406-425
Schilling AG, Möller EM and Geiger HH (1996) PCR-based assays for species-specific detection of Fusarium culmorum, F. graminearum, and F. avenaceum. Phytopathology 86: 515-522
Schnerr H, Vogel RF and Niessen L (2002) Correlation between DNA of trichothecene-producing Fusarium species and deoxynivalenol concentrations in wheat samples. Letters in Applied Microbiology 35: 121-125
Swanson SP, Corley RA, White DG and Buck WB (1984) Rapid thin layer chromatographic method for determination of zearalenone and zearalenol in grains and animal feeds. Journal of Association of Official Analytical Chemists 67: 580-582
Swofford DL (2000) PAUP*: Phylogenetic analysis using parsimony (*and other methods), Version 4b10. Sinauer Associates, Sunderland, Massachusetts, USA
Szécsi A and Bartók T (1995) Trichothecene chemotypes of Fusarium graminearum isolated from corn in Hungary. Mycotoxin Research 11: 85-92
Taylor JW, Jacobson DJ and Fisher MC (1999) The evolution of asexual fungi: Reproduction, speciation and classification. Annual Reviews in Phytopathology 37: 197-246
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F and Higgins DG (1997) The CLUSTAL X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Research 25: 4876-4882
Tibayrenc M (1998) Beyond strain typing and molecular epidemiology: Integrated genetic epidemiology of infectious diseases. Parasitology Today 14: 323-329
Tibayrenc M (1999) Toward an integrated genetic epidemiology of parasitic protozoa and other pathogens. Annual Reviews in Genetics 33: 449-477
Turgeon BG and Yoder OC (2000) Proposed nomenclature for mating type genes of filamentous ascomycetes. Fungal Genetics and Biology 31: 1-5
Trucksess MW, Flood MT, Mossoba MM and Page SW (1987) High-performance thin-layer chromatographic determination of deoxynivalenol, fusarenon-X, and nivalenol in barley, corn, and wheat. Journal of Agricultural and Food Chemistry 35: 444-448
Varga J and Tóth B (2003) Genetic variability and reproductive mode of Aspergillus fumigatus: A review. Infection, Genetics and Evolution 3: 3-17
Ward TJ, Bielawski JP, Kistler HC, Sullivan E and O'Donnell K (2002) Ancestral polymorphism and adaptive evolution in the trichothecene mycotoxin gene cluster of phytopathogenic Fusarium. Proceedings of the National Academy of Sciences USA 99: 9278-9283
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tóth, B., Mesterházy, Á., Nicholson, P. et al. Mycotoxin Production and Molecular Variability of European and American Isolates of Fusarium Culmorum . European Journal of Plant Pathology 110, 587–599 (2004). https://doi.org/10.1023/B:EJPP.0000032398.74570.ab
Issue Date:
DOI: https://doi.org/10.1023/B:EJPP.0000032398.74570.ab